GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
LCK proto-oncogene, Src family tyrosine kinase
Target id: 2053
Nomenclature: LCK proto-oncogene, Src family tyrosine kinase
Abbreviated Name: Lck
Family: Src family
Contents:
- Gene and Protein Information
- Previous and Unofficial Names
- Database Links
- Selected 3D Structures
- Enzyme Reaction
- Inhibitors
- Large-scale Ligand Screening Data
- Immunopharmacology Comments
- Immuno Cell Type Associations
- Immuno Process Associations
- Physiological Functions
- Clinically-Relevant Mutations and Pathophysiology
- References
- How to cite this page
Gene and Protein Information  |
||||||
| Species | TM | AA | Chromosomal Location | Gene Symbol | Gene Name | Reference |
| Human | - | 509 | 1p35.2 | LCK | LCK proto-oncogene, Src family tyrosine kinase | |
| Mouse | - | 509 | 4 63.26 cM | Lck | lck proto-oncogene, Src family tyrosine kinase | |
| Rat | - | 509 | 5q36 | Lck | LCK proto-oncogene, Src family tyrosine kinase | |
Previous and Unofficial Names  |
| Hck-3 | Leukocyte C-terminal Src kinase | p56-LCK | Protein YT16 | lymphocyte-specific protein tyrosine kinase | LCK proto-oncogene |
Database Links  |
|
| Alphafold | P06239 (Hs), P06240 (Mm), Q01621 (Rn) |
| BRENDA | 2.7.10.2 |
| CATH/Gene3D | 3.30.505.10 |
| ChEMBL Target | CHEMBL258 (Hs), CHEMBL2480 (Mm) |
| DrugBank Target | P06239 (Hs) |
| Ensembl Gene | ENSG00000182866 (Hs), ENSMUSG00000000409 (Mm), ENSRNOG00000009705 (Rn) |
| Entrez Gene | 3932 (Hs), 16818 (Mm), 313050 (Rn) |
| Human Protein Atlas | ENSG00000182866 (Hs) |
| KEGG Enzyme | 2.7.10.2 |
| KEGG Gene | hsa:3932 (Hs), mmu:16818 (Mm), rno:313050 (Rn) |
| OMIM | 153390 (Hs) |
| Orphanet | ORPHA280151 (Hs) |
| Pharos | P06239 (Hs) |
| RefSeq Nucleotide | NM_001042771 (Hs), NM_001162432 (Mm), NM_001100709 (Rn) |
| RefSeq Protein | NP_001036236 (Hs), NP_001155904 (Mm), NP_001094179 (Rn) |
| SynPHARM | 84043 (in complex with PP2) |
| UniProtKB | P06239 (Hs), P06240 (Mm), Q01621 (Rn) |
| Wikipedia | LCK (Hs) |
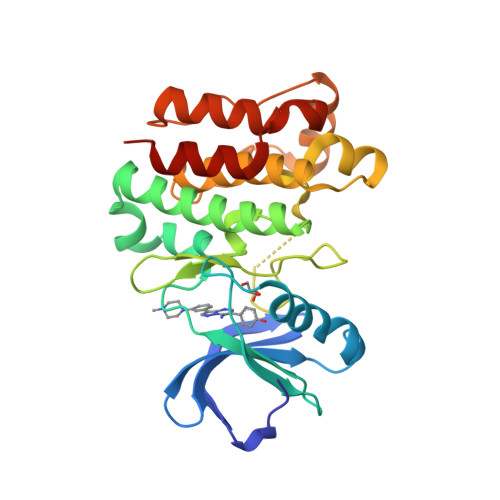
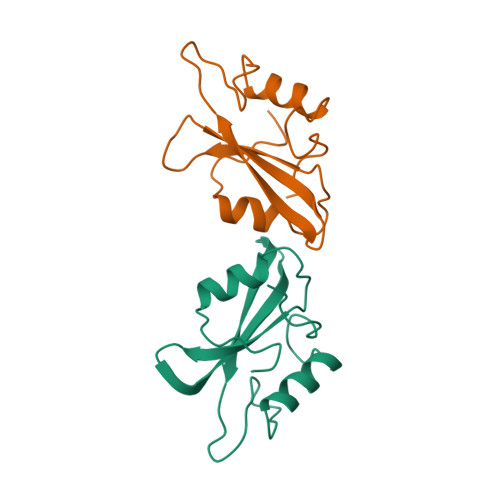
Selected 3D Structures  |
|||||||||||
|
|
||||||||||
|
|
||||||||||
Enzyme Reaction  |
||||
|
||||
Download all structure-activity data for this target as a CSV file 
| Inhibitors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
DiscoveRx KINOMEscan® screen  |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A screen of 72 inhibitors against 456 human kinases. Quantitative data were derived using DiscoveRx KINOMEscan® platform. http://www.discoverx.com/services/drug-discovery-development-services/kinase-profiling/kinomescan Reference: 10,37 |
 
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | Click column headers to sort | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Target used in screen: LCK | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displaying the top 10 most potent ligands View all ligands in screen » | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
EMD Millipore KinaseProfilerTM screen/Reaction Biology Kinase HotspotSM screen  |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A screen profiling 158 kinase inhibitors (Calbiochem Protein Kinase Inhibitor Library I and II, catalogue numbers 539744 and 539745) for their inhibitory activity at 1µM and 10µM against 234 human recombinant kinases using the EMD Millipore KinaseProfilerTM service. A screen profiling the inhibitory activity of 178 commercially available kinase inhibitors at 0.5µM against a panel of 300 recombinant protein kinases using the Reaction Biology Corporation Kinase HotspotSM platform. http://www.millipore.com/techpublications/tech1/pf3036 http://www.reactionbiology.com/webapps/main/pages/kinase.aspx Reference: 1,13 |
 
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Target used in screen: Lck activated | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Target used in screen: Lck/LCK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displaying the top 10 most potent ligands View all ligands in screen » | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunopharmacology Comments |
| Phosphorylation and activation of Lck is an early and critical step in pre-TCR (T cell receptor) and TCR signalling. Activated Lck phosphorylates immunoreceptor tyrosine-based activation motifs of the ζ chain of the TCR leading to recruitment and activation of ZAP-70 tyrosine kinase, and activation of downstream MAPKs and NF-κB. TCR-based signals are required at several stages of T-cell development and it is thought that Lck is the major contributor to TCR signal transduction (with the related Src tyrosine kinase Fyn also playing a role) [29]. JNK pathway-associated phosphatase (JKAP or JSP-1) is reported to dephosphorylate Lck and attenuate TCR signalling [23], with the likelihood that this mechanism would suppressT-cell-mediated immunity and autoimmunity. |
| Cell Type Associations | ||||||
|
||||||
|
| Immuno Process Associations | |||||||||
|
|||||||||
|
|||||||||
|
|||||||||
|
|||||||||
|
|||||||||
|
| Physiological Functions Comments | |
| Lck is required for the T cell receptor (TCR)-mediated signalling underlying normal T cell development and activation [15,21,32-33]. Selective inhibition of Lck offers a new strategy for the treatment of graft rejection and/or T cell-mediated autoimmune and inflammatory disease (eg rheumatoid arthritis, psoriasis, multiple sclerosis, atherosclerosis). |
Clinically-Relevant Mutations and Pathophysiology 
|
||||||||||||
|
||||||||||||
References
1. Anastassiadis T, Deacon SW, Devarajan K, Ma H, Peterson JR. (2011) Comprehensive assay of kinase catalytic activity reveals features of kinase inhibitor selectivity. Nat Biotechnol, 29 (11): 1039-45. [PMID:22037377]
2. Bain J, Plater L, Elliott M, Shpiro N, Hastie CJ, McLauchlan H, Klevernic I, Arthur JS, Alessi DR, Cohen P. (2007) The selectivity of protein kinase inhibitors: a further update. Biochem J, 408 (3): 297-315. [PMID:17850214]
3. Bamborough P, Angell RM, Bhamra I, Brown D, Bull J, Christopher JA, Cooper AW, Fazal LH, Giordano I, Hind L et al.. (2007) N-4-Pyrimidinyl-1H-indazol-4-amine inhibitors of Lck: indazoles as phenol isosteres with improved pharmacokinetics. Bioorg Med Chem Lett, 17 (15): 4363-8. [PMID:17600705]
4. Barf TA, Jans CGJM, Man PADeA, Oubrie AA, Raaijmakers HCA, Rewinkel JBM, Sterrenburg J-G, Wijkmans JCHM. (2014) 4-imidazopyridazin-1-yl-benzamides and 4-imidazotriazin-1-yl-benzamides as btk inhibitors. Patent number: US20140155385 A1. Assignee: Barf TA, Jans CGJM, Man PADeA, Oubrie AA, Raaijmakers HCA, Rewinkel JBM, Sterrenburg J-G, Wijkmans JCHM. Priority date: 19/07/2011. Publication date: 05/06/2014.
5. Barker MD, Liddle J, Atkinson FL, Wilson DM, Dickson MC, Ramirez-Molina C, Lewis H, Davis RP, Somers DO, Neu M et al.. (2018) Discovery of potent and selective Spleen Tyrosine Kinase inhibitors for the topical treatment of inflammatory skin disease. Bioorg Med Chem Lett, 28 (21): 3458-3462. [PMID:30249354]
6. Burchat AF, Calderwood DJ, Hirst GC, Holman NJ, Johnston DN, Munschauer R, Rafferty P, Tometzki GB. (2000) Pyrrolo[2,3-d]pyrimidines containing an extended 5-substituent as potent and selective inhibitors of lck II. Bioorg Med Chem Lett, 10 (19): 2171-4. [PMID:11012022]
7. Byrd JC, Harrington B, O'Brien S, Jones JA, Schuh A, Devereux S, Chaves J, Wierda WG, Awan FT, Brown JR et al.. (2016) Acalabrutinib (ACP-196) in Relapsed Chronic Lymphocytic Leukemia. N Engl J Med, 374 (4): 323-32. [PMID:26641137]
8. Castro-Falcón G, Seiler GS, Demir Ö, Rathinaswamy MK, Hamelin D, Hoffmann RM, Makowski SL, Letzel AC, Field SJ, Burke JE et al.. (2018) Neolymphostin A Is a Covalent Phosphoinositide 3-Kinase (PI3K)/Mammalian Target of Rapamycin (mTOR) Dual Inhibitor That Employs an Unusual Electrophilic Vinylogous Ester. J Med Chem, 61 (23): 10463-10472. [PMID:30380865]
9. Chen P, Norris D, Das J, Spergel SH, Wityak J, Leith L, Zhao R, Chen BC, Pitt S, Pang S et al.. (2004) Discovery of novel 2-(aminoheteroaryl)-thiazole-5-carboxamides as potent and orally active Src-family kinase p56(Lck) inhibitors. Bioorg Med Chem Lett, 14 (24): 6061-6. [PMID:15546730]
10. Davis MI, Hunt JP, Herrgard S, Ciceri P, Wodicka LM, Pallares G, Hocker M, Treiber DK, Zarrinkar PP. (2011) Comprehensive analysis of kinase inhibitor selectivity. Nat Biotechnol, 29 (11): 1046-51. [PMID:22037378]
11. DiMauro EF, Newcomb J, Nunes JJ, Bemis JE, Boucher C, Buchanan JL, Buckner WH, Cheng A, Faust T, Hsieh F et al.. (2007) Discovery of 4-amino-5,6-biaryl-furo[2,3-d]pyrimidines as inhibitors of Lck: development of an expedient and divergent synthetic route and preliminary SAR. Bioorg Med Chem Lett, 17 (8): 2305-9. [PMID:17280833]
12. Fraser C, Dawson JC, Dowling R, Houston DR, Weiss JT, Munro AF, Muir M, Harrington L, Webster SP, Frame MC et al.. (2016) Rapid Discovery and Structure-Activity Relationships of Pyrazolopyrimidines That Potently Suppress Breast Cancer Cell Growth via SRC Kinase Inhibition with Exceptional Selectivity over ABL Kinase. J Med Chem, 59 (10): 4697-710. [PMID:27115835]
13. Gao Y, Davies SP, Augustin M, Woodward A, Patel UA, Kovelman R, Harvey KJ. (2013) A broad activity screen in support of a chemogenomic map for kinase signalling research and drug discovery. Biochem J, 451 (2): 313-28. [PMID:23398362]
14. Girotti MR, Lopes F, Preece N, Niculescu-Duvaz D, Zambon A, Davies L, Whittaker S, Saturno G, Viros A, Pedersen M et al.. (2015) Paradox-breaking RAF inhibitors that also target SRC are effective in drug-resistant BRAF mutant melanoma. Cancer Cell, 27 (1): 85-96. [PMID:25500121]
15. Goldman FD, Ballas ZK, Schutte BC, Kemp J, Hollenback C, Noraz N, Taylor N. (1998) Defective expression of p56lck in an infant with severe combined immunodeficiency. J Clin Invest, 102 (2): 421-9. [PMID:9664084]
16. Gommermann N, Buehlmayer P, von Matt A, Breitenstein W, Masuya K, Pirard B, Furet P, Cowan-Jacob SW, Weckbecker G. (2010) New pyrazolo[1,5a]pyrimidines as orally active inhibitors of Lck. Bioorg Med Chem Lett, 20 (12): 3628-31. [PMID:20483608]
17. Hanke JH, Gardner JP, Dow RL, Changelian PS, Brissette WH, Weringer EJ, Pollok BA, Connelly PA. (1996) Discovery of a novel, potent, and Src family-selective tyrosine kinase inhibitor. Study of Lck- and FynT-dependent T cell activation. J Biol Chem, 271 (2): 695-701. [PMID:8557675]
18. Hennequin LF, Allen J, Breed J, Curwen J, Fennell M, Green TP, Lambert-van der Brempt C, Morgentin R, Norman RA, Olivier A et al.. (2006) N-(5-chloro-1,3-benzodioxol-4-yl)-7-[2-(4-methylpiperazin-1-yl)ethoxy]-5- (tetrahydro-2H-pyran-4-yloxy)quinazolin-4-amine, a novel, highly selective, orally available, dual-specific c-Src/Abl kinase inhibitor. J Med Chem, 49 (22): 6465-88. [PMID:17064066]
19. Jiang X, Zhao B, Britton R, Lim LY, Leong D, Sanghera JS, Zhou BB, Piers E, Andersen RJ, Roberge M. (2004) Inhibition of Chk1 by the G2 DNA damage checkpoint inhibitor isogranulatimide. Mol Cancer Ther, 3 (10): 1221-7. [PMID:15486189]
20. Kim KH, Maderna A, Schnute ME, Hegen M, Mohan S, Miyashiro J, Lin L, Li E, Keegan S, Lussier J et al.. (2011) Imidazo[1,5-a]quinoxalines as irreversible BTK inhibitors for the treatment of rheumatoid arthritis. Bioorg Med Chem Lett, 21 (21): 6258-63. [PMID:21958547]
21. Levin SD, Anderson SJ, Forbush KA, Perlmutter RM. (1993) A dominant-negative transgene defines a role for p56lck in thymopoiesis. EMBO J, 12 (4): 1671-80. [PMID:8385609]
22. Lewis RT, Bode CM, Choquette DM, Potashman M, Romero K, Stellwagen JC, Teffera Y, Moore E, Whittington DA, Chen H et al.. (2012) The discovery and optimization of a novel class of potent, selective, and orally bioavailable anaplastic lymphoma kinase (ALK) inhibitors with potential utility for the treatment of cancer. J Med Chem, 55 (14): 6523-40. [PMID:22734674]
23. Li JP, Yang CY, Chuang HC, Lan JL, Chen DY, Chen YM, Wang X, Chen AJ, Belmont JW, Tan TH. (2014) The phosphatase JKAP/DUSP22 inhibits T-cell receptor signalling and autoimmunity by inactivating Lck. Nat Commun, 5: 3618. [PMID:24714587]
24. Lowell CA. (2004) Src-family kinases: rheostats of immune cell signaling. Mol Immunol, 41 (6-7): 631-43. [PMID:15220000]
25. Machrouhi F, Ouhamou N, Laderoute K, Calaoagan J, Bukhtiyarova M, Ehrlich PJ, Klon AE. (2010) The rational design of a novel potent analogue of the 5'-AMP-activated protein kinase inhibitor compound C with improved selectivity and cellular activity. Bioorg Med Chem Lett, 20 (22): 6394-9. [PMID:20932747]
26. Manthey CL, Johnson DL, Illig CR, Tuman RW, Zhou Z, Baker JF, Chaikin MA, Donatelli RR, Franks CF, Zeng L et al.. (2009) JNJ-28312141, a novel orally active colony-stimulating factor-1 receptor/FMS-related receptor tyrosine kinase-3 receptor tyrosine kinase inhibitor with potential utility in solid tumors, bone metastases, and acute myeloid leukemia. Mol Cancer Ther, 8 (11): 3151-61. [PMID:19887542]
27. Martin MW, Newcomb J, Nunes JJ, McGowan DC, Armistead DM, Boucher C, Buchanan JL, Buckner W, Chai L, Elbaum D et al.. (2006) Novel 2-aminopyrimidine carbamates as potent and orally active inhibitors of Lck: synthesis, SAR, and in vivo antiinflammatory activity. J Med Chem, 49 (16): 4981-91. [PMID:16884310]
28. McLean LR, Zhang Y, Zaidi N, Bi X, Wang R, Dharanipragada R, Jurcak JG, Gillespy TA, Zhao Z, Musick KY et al.. (2012) X-ray crystallographic structure-based design of selective thienopyrazole inhibitors for interleukin-2-inducible tyrosine kinase. Bioorg Med Chem Lett, 22 (9): 3296-300. [PMID:22464456]
29. Palacios EH, Weiss A. (2004) Function of the Src-family kinases, Lck and Fyn, in T-cell development and activation. Oncogene, 23 (48): 7990-8000. [PMID:15489916]
30. Ren Y, Zheng J, Fan S, Wang L, Cheng M, Shi D, Zhang W, Tang R, Yu Y, Jiao L et al.. (2017) Anti-tumor efficacy of theliatinib in esophageal cancer patient-derived xenografts models with epidermal growth factor receptor (EGFR) overexpression and gene amplification. Oncotarget, 8 (31): 50832-50844. [PMID:28881608]
31. Sabat M, Dougan DR, Knight B, Lawson JD, Scorah N, Smith CR, Taylor ER, Vu P, Wyrick C, Wang H et al.. (2021) Discovery of the Bruton's Tyrosine Kinase Inhibitor Clinical Candidate TAK-020 (S)-5-(1-((1-Acryloylpyrrolidin-3-yl)oxy)isoquinolin-3-yl)-2,4-dihydro-3H-1,2,4-triazol-3-one, by Fragment-Based Drug Design. J Med Chem, 64 (17): 12893-12902. [PMID:34448571]
32. Seddon B, Zamoyska R. (2002) TCR signals mediated by Src family kinases are essential for the survival of naive T cells. J Immunol, 169 (6): 2997-3005. [PMID:12218114]
33. Straus DB, Weiss A. (1992) Genetic evidence for the involvement of the lck tyrosine kinase in signal transduction through the T cell antigen receptor. Cell, 70 (4): 585-93. [PMID:1505025]
34. Tap WD, Wainberg ZA, Anthony SP, Ibrahim PN, Zhang C, Healey JH, Chmielowski B, Staddon AP, Cohn AL, Shapiro GI et al.. (2015) Structure-Guided Blockade of CSF1R Kinase in Tenosynovial Giant-Cell Tumor. N Engl J Med, 373 (5): 428-37. [PMID:26222558]
35. Tong L, Warren TC, Lukas S, Schembri-King J, Betageri R, Proudfoot JR, Jakes S. (1998) Carboxymethyl-phenylalanine as a replacement for phosphotyrosine in SH2 domain binding. J Biol Chem, 273 (32): 20238-42. [PMID:9685372]
36. Wang Z, Zhang Y, Pinkas DM, Fox AE, Luo J, Huang H, Cui S, Xiang Q, Xu T, Xun Q et al.. (2018) Design, Synthesis, and Biological Evaluation of 3-(Imidazo[1,2- a]pyrazin-3-ylethynyl)-4-isopropyl- N-(3-((4-methylpiperazin-1-yl)methyl)-5-(trifluoromethyl)phenyl)benzamide as a Dual Inhibitor of Discoidin Domain Receptors 1 and 2. J Med Chem, 61 (17): 7977-7990. [PMID:30075624]
37. Wodicka LM, Ciceri P, Davis MI, Hunt JP, Floyd M, Salerno S, Hua XH, Ford JM, Armstrong RC, Zarrinkar PP et al.. (2010) Activation state-dependent binding of small molecule kinase inhibitors: structural insights from biochemistry. Chem Biol, 17 (11): 1241-9. [PMID:21095574]
38. Xu Z, Zhang N, Wang T, Sun Q, Wang Y. (2017) Fused pyrimidine compound, and intermediate, preparation method, composition and application of. Patent number: CN106366093A. Assignee: Guangzhou Re Pharmaceutical Technology Co Ltd. Priority date: 21/07/2016. Publication date: 01/02/2017.
39. Yang J, Shibu MA, Kong L, Luo J, BadrealamKhan F, Huang Y, Tu ZC, Yun CH, Huang CY, Ding K et al.. (2020) Design, Synthesis, and Structure-Activity Relationships of 1,2,3-Triazole Benzenesulfonamides as New Selective Leucine-Zipper and Sterile-α Motif Kinase (ZAK) Inhibitors. J Med Chem, 63 (5): 2114-2130. [PMID:31244114]
40. Zeng H, Belanger DB, Curran PJ, Shipps Jr GW, Miao H, Bracken JB, Arshad Siddiqui M, Malkowski M, Wang Y. (2011) Discovery of novel imidazo[1,2-a]pyrazin-8-amines as Brk/PTK6 inhibitors. Bioorg Med Chem Lett, 21 (19): 5870-5. [PMID:21855335]
How to cite this page
Src family: LCK proto-oncogene, Src family tyrosine kinase. Last modified on 06/03/2024. Accessed on 20/05/2026. IUPHAR/BPS Guide to PHARMACOLOGY, https://www.guidetomalariapharmacology.org/GRAC/ObjectDisplayForward?objectId=2053.