|
Synonyms: ABDNAZ [2] | RRx-001 | RRx001
Compound class:
Synthetic organic
Comment: RRx-001 (EpicentRx) was originally developed as an anti-tumour agent [3]; it promotes apoptosis, and has radio- , chemo- and immuno-sensitising actions in cancer models. It also provides cytoprotective effects in normal tissues, which has led to the proposal of RRx-001 as a supportive care drug in patients undergoing cytotoxic cancer therapy. RRx-001 was identified using phenotypic screening, so its molecular mechanism of action was initially unclear. Several mechanisms have subsequently been identified, including NLRP3 inflammasome inhibition [1,4] and CD47/SIRP-α immune checkpoint inhibition. RRx-001 covalently targets the cysteine 409 residue in NLRP3's NACHT domain and blocks NLRP3/NEK7 interaction and assembly of the inflammasome complex. This ultimately inhibits release of active pro-inflammatory cytokines (IL-1β and IL-18).
RRx-001's chemical structure is a match for the INN nibrozetone (proposed INN list 130 of Feb. 2024). |
|
|||||||||||||||||||||||||||||||||||
Classification  |
|
| Compound class | Synthetic organic |
IUPAC Name  |
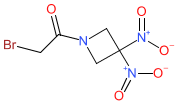
| 2-bromo-1-(3,3-dinitroazetidin-1-yl)ethanone |
International Nonproprietary Names  |
|
| INN number | INN |
| 12836 | nibrozetone |
Synonyms  |
|
| ABDNAZ [2] | RRx-001 | RRx001 | |
Database Links  |
|
| ChEMBL Ligand | CHEMBL3526802 |
| DrugBank Ligand | DB12060 |
| GtoPdb PubChem SID | 491299966 |
| PubChem CID | 15950826 |
| Search Google for chemical match using the InChIKey | JODKFOVZURLVTG-UHFFFAOYSA-N |
| Search Google for chemicals with the same backbone | JODKFOVZURLVTG |
| Search PubMed clinical trials | nibrozetone |
| Search PubMed titles | nibrozetone |
| Search PubMed titles/abstracts | nibrozetone |
| UniChem Compound Search for chemical match using the InChIKey | JODKFOVZURLVTG-UHFFFAOYSA-N |
| UniChem Connectivity Search for chemical match using the InChIKey | JODKFOVZURLVTG-UHFFFAOYSA-N |