GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
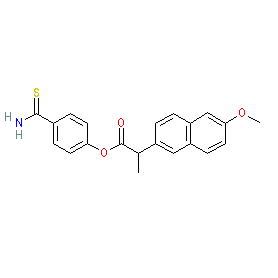
Synonyms: ATB 346 | ATB-346 | ATB-346, (S)- | ATB346
Compound class:
Synthetic organic
Comment: Otenaproxesul (ATB-346) is a novel, orally administered, gastrointestinal-sparing hydrogen sulfide (H2S)-releasing analgesic and anti-inflammatory drug in clinical trials for osteoarthritis [8]. Structurally it comprises an H2S-releasing moiety covalently linked to a NSAID (naproxen). H2S-releasing NSAIDs are in development for their potential analgesic, anti-inflammatory, cytoprotective and chemopreventative properties [2-3,5-6,9-10].
|
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Blackler R, Syer S, Bolla M, Ongini E, Wallace JL. (2012)
Gastrointestinal-sparing effects of novel NSAIDs in rats with compromised mucosal defence. PLoS ONE, 7 (4): e35196. [PMID:22496907] |
|
2. De Cicco P, Panza E, Ercolano G, Armogida C, Sessa G, Pirozzi G, Cirino G, Wallace JL, Ianaro A. (2016)
ATB-346, a novel hydrogen sulfide-releasing anti-inflammatory drug, induces apoptosis of human melanoma cells and inhibits melanoma development in vivo. Pharmacol Res, 114: 67-73. [PMID:27777130] |
|
3. Elsheikh W, Blackler RW, Flannigan KL, Wallace JL. (2014)
Enhanced chemopreventive effects of a hydrogen sulfide-releasing anti-inflammatory drug (ATB-346) in experimental colorectal cancer. Nitric Oxide, 41: 131-7. [PMID:24747869] |
|
4. Fomenko I, Sklyarov A, Bondarchuk T, Biletska L, Panasyuk N, Wallace JL. (2014)
Effects of conventional and hydrogen sulfide-releasing non-steroidal anti-inflammatory drugs in rats with stress-induced and epinephrine-induced gastric damage. Stress, 17 (6): 528-37. [PMID:25238023] |
|
5. Gemici B, Elsheikh W, Feitosa KB, Costa SK, Muscara MN, Wallace JL. (2015)
H2S-releasing drugs: anti-inflammatory, cytoprotective and chemopreventative potential. Nitric Oxide, 46: 25-31. [PMID:25461269] |
|
6. Szabo C, Papapetropoulos A. (2017)
International Union of Basic and Clinical Pharmacology. CII: Pharmacological Modulation of H2S Levels: H2S Donors and H2S Biosynthesis Inhibitors. Pharmacol Rev, 69 (4): 497-564. [PMID:28978633] |
|
7. Wallace JL, Caliendo G, Santagada V, Cirino G. (2010)
Markedly reduced toxicity of a hydrogen sulphide-releasing derivative of naproxen (ATB-346). Br J Pharmacol, 159 (6): 1236-46. [PMID:20128814] |
|
8. Wallace JL, Nagy P, Feener TD, Allain T, Ditrói T, Vaughan DJ, Muscara MN, de Nucci G, Buret AG. (2020)
A proof-of-concept, Phase 2 clinical trial of the gastrointestinal safety of a hydrogen sulfide-releasing anti-inflammatory drug. Br J Pharmacol, 177 (4): 769-777. [PMID:30834513] |
|
9. Wallace JL, Vaughan D, Dicay M, MacNaughton WK, de Nucci G. (2018)
Hydrogen Sulfide-Releasing Therapeutics: Translation to the Clinic. Antioxid Redox Signal, 28 (16): 1533-1540. [PMID:28388861] |
|
10. Zheng Y, Ji X, Ji K, Wang B. (2015)
Hydrogen sulfide prodrugs-a review. Acta Pharm Sin B, 5 (5): 367-77. [PMID:26579468] |