GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
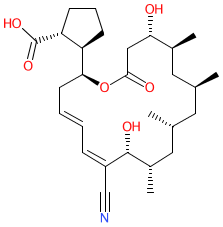
Synonyms: (-)-borrelidin
Compound class:
Natural product
Comment: Borrelidin is a macrolide antibacterial originally isolated from Streptomyces. It is a noncompetitive inhibitor of both bacterial and eukaryotic threonyl-tRNA synthetases (also known as threonine--tRNA ligase), exhibiting antibacterial, antimalarial and antiangiogenic activities.
The Malaria tab on this ligand page provides additional curator comments of relevance to the Guide to MALARIA PHARMACOLOGY Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Novoa EM, Camacho N, Tor A, Wilkinson B, Moss S, Marín-García P, Azcárate IG, Bautista JM, Mirando AC, Francklyn CS et al.. (2014)
Analogs of natural aminoacyl-tRNA synthetase inhibitors clear malaria in vivo. Proc Natl Acad Sci U S A, 111 (51): E5508-17. [PMID:25489076] |
|
2. Otoguro K, Ui H, Ishiyama A, Kobayashi M, Togashi H, Takahashi Y, Masuma R, Tanaka H, Tomoda H, Yamada H et al.. (2003)
In vitro and in vivo antimalarial activities of a non-glycosidic 18-membered macrolide antibiotic, borrelidin, against drug-resistant strains of Plasmodia. J Antibiot (Tokyo), 56 (8): 727-9. [PMID:14563165] |
|
3. Sugawara A, Tanaka T, Hirose T, Ishiyama A, Iwatsuki M, Takahashi Y, Otoguro K, Ōmura S, Sunazuka T. (2013)
Borrelidin analogues with antimalarial activity: design, synthesis and biological evaluation against Plasmodium falciparum parasites. Bioorg Med Chem Lett, 23 (8): 2302-5. [PMID:23499502] |