GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Synonyms: EIDD-2801 | EIDD2801 | Lagevrio® | MK-4482 | MK4482
molnupiravir is an approved drug (UK (2021))
Compound class:
Synthetic organic
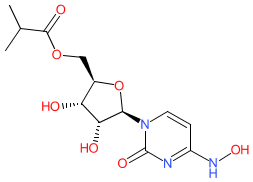
Comment: Molnupiravir (EIDD-2801, MK-4482) is an oral, broad-spectrum antiviral drug [1,8]. The proposed INN molnupiravir was released by the WHO in a special release to cover COVID-related therapeutics in October 2020 (Proposed INN: List 124-COVID-19). Chemically molnupiravir is the isopropylester prodrug of the ribonucleoside analogue β-D-N4-hydroxycytidine (EIDD-1931, or N-hydroxycytidine) [10-11]. Functionally, the active form of molnupiravir is incorporated into the virus' RNA by the viral RNA-dependent RNA polymerase (RdRp) during replication, which results in lethal mutagenesis [4]. Molnupiravir was originally designed by Emory University scientists to inhibit replication of influenza virus. It has subsequently been tested for activity against other RNA viruses, including pandemic SARS-CoV-2, SARS-CoV and MERS-CoV [14]. Molnupiravir was being progressed for COVID-19 in a collaboration between Ridgeback Biotherapeutics and Merck. In vivo therapeutic and prophylactic potential was reported in a peer reviewed manuscript in February 2021 [12].
There is concern that molnupiravir may induce mutations in the host, based on evidence from mammalian cell culture experiments [13] and using the Ames test (bacteria). |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Bimonte S, Crispo A, Amore A, Celentano E, Cuomo A, Cascella M. (2020)
Potential Antiviral Drugs for SARS-Cov-2 Treatment: Preclinical Findings and Ongoing Clinical Research. In Vivo, 34 (3 Suppl): 1597-1602. [PMID:32503817] |
|
2. Eddy J.
Ridgeback Biotherapeutics Announces Launch of Phase 2 Trials Testing EIDD-2801 as Potential Treatment for COVID-19. Accessed on 01/07/2020. Modified on 01/07/2020. businesswire.com, https://www.businesswire.com/news/home/20200619005038/en/Ridgeback-Biotherapeutics-Announces-Launch-Phase-2-Trials |
|
3. Jayk Bernal A, Gomes da Silva MM, Musungaie DB, Kovalchuk E, Gonzalez A, Delos Reyes V, Martín-Quirós A, Caraco Y, Williams-Diaz A, Brown ML et al.. (2022)
Molnupiravir for Oral Treatment of Covid-19 in Nonhospitalized Patients. N Engl J Med, 386 (6): 509-520. [PMID:34914868] |
|
4. Kabinger F, Stiller C, Schmitzová J, Dienemann C, Kokic G, Hillen HS, Höbartner C, Cramer P. (2021)
Mechanism of molnupiravir-induced SARS-CoV-2 mutagenesis. Nat Struct Mol Biol, 28 (9): 740-746. [PMID:34381216] |
|
5. Khoo SH, FitzGerald R, Saunders G, Middleton C, Ahmad S, Edwards CJ, Hadjiyiannakis D, Walker L, Lyon R, Shaw V et al.. (2022)
Molnupiravir versus placebo in unvaccinated and vaccinated patients with early SARS-CoV-2 infection in the UK (AGILE CST-2): a randomised, placebo-controlled, double-blind, phase 2 trial. The Lancet, Epub ahead of print. DOI: 10.1016/S1473-3099(22)00644-2 |
|
6. Merck.
Merck and Ridgeback’s Investigational Oral Antiviral Molnupiravir Reduced the Risk of Hospitalization or Death by Approximately 50 Percent Compared to Placebo for Patients with Mild or Moderate COVID-19 in Positive Interim Analysis of Phase 3 Study. Accessed on 01/10/2021. Modified on 01/10/2021. www.merck.com/news/, https://www.merck.com/news/merck-and-ridgebacks-investigational-oral-antiviral-molnupiravir-reduced-the-risk-of-hospitalization-or-death-by-approximately-50-percent-compared-to-placebo-for-patients-with-mild-or-moderat/ |
|
7. Mollan KR, Eron JJ, Krajewski TJ, Painter W, Duke ER, Morse CG, Goecker EA, Premkumar L, Wolfe CR, Szewczyk LJ et al.. (2022)
Infectious Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Virus in Symptomatic Coronavirus Disease 2019 (COVID-19) Outpatients: Host, Disease, and Viral Correlates. Clin Infect Dis, 75 (1): e1028-e1036 Preprint. DOI: 10.1093/cid/ciab968 [PMID:35022711] |
|
8. Sheahan TP, Sims AC, Zhou S, Graham RL, Pruijssers AJ, Agostini ML, Leist SR, Schäfer A, Dinnon 3rd KH, Stevens LJ et al.. (2020)
An orally bioavailable broad-spectrum antiviral inhibits SARS-CoV-2 in human airway epithelial cell cultures and multiple coronaviruses in mice. Sci Transl Med, 12 (541). [PMID:32253226] |
|
9. Syed YY. (2022)
Molnupiravir: First Approval. Drugs, 82 (4): 455-460. [PMID:35184266] |
|
10. Toots M, Yoon JJ, Cox RM, Hart M, Sticher ZM, Makhsous N, Plesker R, Barrena AH, Reddy PG, Mitchell DG et al.. (2019)
Characterization of orally efficacious influenza drug with high resistance barrier in ferrets and human airway epithelia. Sci Transl Med, 11 (515). DOI: 10.1126/scitranslmed.aax5866 [PMID:31645453] |
|
11. Toots M, Yoon JJ, Hart M, Natchus MG, Painter GR, Plemper RK. (2020)
Quantitative efficacy paradigms of the influenza clinical drug candidate EIDD-2801 in the ferret model. Transl Res, 218: 16-28. [PMID:31945316] |
|
12. Wahl A, Gralinski LE, Johnson CE, Yao W, Kovarova M, Dinnon 3rd KH, Liu H, Madden VJ, Krzystek HM, De C et al.. (2021)
SARS-CoV-2 infection is effectively treated and prevented by EIDD-2801. Nature, 591 (7850): 451-457. [PMID:33561864] |
|
13. Zhou S, Hill CS, Sarkar S, Tse LV, Woodburn BMD, Schinazi RF, Sheahan TP, Baric RS, Heise MT, Swanstrom R. (2021)
β-d-N4-hydroxycytidine Inhibits SARS-CoV-2 Through Lethal Mutagenesis But Is Also Mutagenic To Mammalian Cells. J Infect Dis, 224 (3): 415-419. [PMID:33961695] |
|
14. (1988)
Bombesin-like peptides in health and disease. Proceedings of an international symposium. October 13-16, 1987, Rome, Italy. A tribute to Vittorio Erspamer, M.D. Ann NY Acad Sci, 547: 1-541. [PMID:3071214] |