GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Synonyms: EIDD-2801 | EIDD2801 | Lagevrio® | MK-4482 | MK4482
molnupiravir is an approved drug (UK (2021))
Compound class:
Synthetic organic
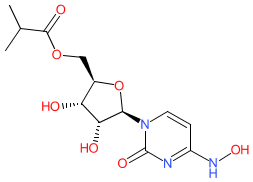
Comment: Molnupiravir (EIDD-2801, MK-4482) is an oral, broad-spectrum antiviral drug [1,8]. The proposed INN molnupiravir was released by the WHO in a special release to cover COVID-related therapeutics in October 2020 (Proposed INN: List 124-COVID-19). Chemically molnupiravir is the isopropylester prodrug of the ribonucleoside analogue β-D-N4-hydroxycytidine (EIDD-1931, or N-hydroxycytidine) [10-11]. Functionally, the active form of molnupiravir is incorporated into the virus' RNA by the viral RNA-dependent RNA polymerase (RdRp) during replication, which results in lethal mutagenesis [4]. Molnupiravir was originally designed by Emory University scientists to inhibit replication of influenza virus. It has subsequently been tested for activity against other RNA viruses, including pandemic SARS-CoV-2, SARS-CoV and MERS-CoV [14]. Molnupiravir was being progressed for COVID-19 in a collaboration between Ridgeback Biotherapeutics and Merck. In vivo therapeutic and prophylactic potential was reported in a peer reviewed manuscript in February 2021 [12].
There is concern that molnupiravir may induce mutations in the host, based on evidence from mammalian cell culture experiments [13] and using the Ames test (bacteria). |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| A first-in-human safety, tolerability and pharmacokinetics RCT investigating molnupiravir/EIDD-2801 (in healthy volunteers) in the UK began in mid-April 2020 and recruitment of US participants began a few weeks later. Although Phase 1 safety results have not yet been published, Ridgeback Biotherapeutics have announced via press release (June 19, 2020) that EIDD-2801 is safe in humans at predicted therapeutic doses. At this time Ridgeback advised that they would launch two Phase 2 clinical trials in recently symptomatic, newly diagnosed COVID-19 patients (separate trials for newly hospitalised and newly diagnosed non-hospitalised patient groups) [2]. These studies recruited participants in the US and in the UK. In April 2021, Merck announced that they were discontinuing development of molnupiravir in hospitalised patients after a predicted lack of efficacy became evident from the clinical trial. In contrast a company press release from Merck on 1st October 2021 [6], revealed that so significant was the clinical benefit of molnupiravir in at-risk, non-hospitalized adult patients with mild-to-moderate COVID-19, the FDA Data and Safety Monitoring Board recommended stopping the trial early, viewing it unethical to proceed. At a planned interim analysis of data collected from an initial set of >750 participants (recruited between May and early August), molnupiravir reduced the risk of hospitalisation or death by ~50% in patients with confirmed mild-to-moderate COVID-19 and at least one risk factor associated with poor disease outcome (on a regimen of 4 tablets of drug or placebo, twice daily for 5 consecutive days). However, treatment in a subsequent cohort of ~650 participants (recruited between August and early October) was found to be less effective than previously thought. In response to the original efficacy data, Ridgeback/Merck proceeded to submit applications for approval to various national drug regulatory agencies. Molnupiravir's first approval was issued by the UK's MHRA on 4th Nov. 2021, as a treatment for recently diagnosed SARS-CoV-2 infection in non-hospitalised individuals who have at least one risk factor for developing severe illness [9]. The US FDA recommended molnupiravir for EUA in late November 2021. Approvals by other national regulatory agencies are pending. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT04392219 | COVID-19 First In Human Study to Evaluate Safety, Tolerability, and Pharmacokinetics of EIDD-2801 in Healthy Volunteers | Phase 1 Interventional | Ridgeback Biotherapeutics, LP | ||
| NCT04405739 | The Effect of EIDD-2801 on Viral Shedding of SARS-CoV-2 (COVID-19) | Phase 2 Interventional | Ridgeback Biotherapeutics, LP | ||
| NCT04405570 | A Safety, Tolerability and Efficacy of EIDD-2801 to Eliminate Infectious Virus Detection in Persons With COVID-19 | Phase 2 Interventional | Ridgeback Biotherapeutics, LP | 7 | |
| NCT04575584 | Efficacy and Safety of Molnupiravir (MK-4482) in Hospitalized Adult Participants With COVID-19 (MK-4482-001) | Phase 2/Phase 3 Interventional | Merck Sharp & Dohme Corp. | ||
| NCT04575597 | Efficacy and Safety of Molnupiravir (MK-4482) in Non-Hospitalized Adult Participants With COVID-19 (MK-4482-002) | Phase 2/Phase 3 Interventional | Merck Sharp & Dohme Corp. | MOVe-OUT trial: A press release from Merck in Oct 2021 indicated that at a planned interim analysis molnupiravir reduced the risk of hospitalisation or death by approximately 50% in patients with confirmed mild-to-moderate COVID-19 and at least one risk factor associated with poor disease outcome. The clinical benefit was evident across viral variants such as Gamma, Delta, and Mu. | 3 |
| NCT04746183 | AGILE (Early Phase Platform Trial for COVID-19) | Phase 1/Phase 2 Interventional | University of Liverpool | This is the AGILE CST-2 study that was carried out in the UK between November 2020 and March 2022. | 5 |
External links  |
|
For extended ADME data see the following: Electronic Medicines Compendium (eMC) Drugs.com |