GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Synonyms: 7351 | Malaridine®
Compound class:
Synthetic organic
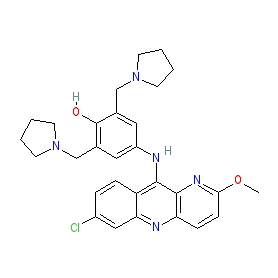
Comment: Pyronaridine is a benzonaphthyridine derivative with potent antimalarial activity.
The Malaria tab on this ligand page provides additional curator comments of relevance to the Guide to MALARIA PHARMACOLOGY. |
|
|||||||||||||||||||||||||||||||||||
| Guide to Malaria Pharmacology Comments |
| Pyronaridine was synthesized in the early 1970s in China (as part of Project 523) and used in that jurisdiction for the treatment of malaria as a monotherapy. Clinical data show excellent antimalarial efficacy against P. falciparum (including chloroquine resistant strains) and combination therapy studies also show promise against P. vivax [1]. Pyronaridine is listed in the World Health Organisation's 21st Essential Medicines List (2019) as an antimalarial for curative treatment in combination with artesunate (trade name Pyramax®). This is one of the new artemisinin-based combination therapies (ACTs) registered and used in some countries but not yet recommended by the WHO for unrestricted use [3]. |