GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Synonyms: 7351 | Malaridine®
Compound class:
Synthetic organic
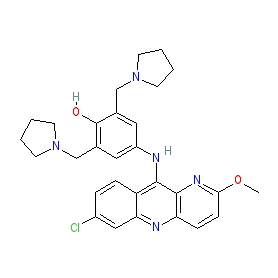
Comment: Pyronaridine is a benzonaphthyridine derivative with potent antimalarial activity.
The Malaria tab on this ligand page provides additional curator comments of relevance to the Guide to MALARIA PHARMACOLOGY. |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Croft SL, Duparc S, Arbe-Barnes SJ, Craft JC, Shin CS, Fleckenstein L, Borghini-Fuhrer I, Rim HJ. (2012)
Review of pyronaridine anti-malarial properties and product characteristics. Malar J, 11: 270. [PMID:22877082] |
|
2. Delves M, Plouffe D, Scheurer C, Meister S, Wittlin S, Winzeler EA, Sinden RE, Leroy D. (2012)
The activities of current antimalarial drugs on the life cycle stages of Plasmodium: a comparative study with human and rodent parasites. PLoS Med, 9 (2): e1001169. [PMID:22363211] |
|
3. (2015)
All chapters. In Guidelines for the Treatment of Malaria (World Health Organization) . [PMID:26020088] [ISBN:9789241549127] |