GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
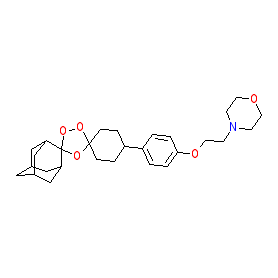
Synonyms: OZ439
Compound class:
Synthetic organic
Comment: Artefenomel is a synthetic ozonide (synthetic peroxide), a class of compound that retains the endoperoxide bridge of the artemisinin class of antimalarials but also contains the sterically bulky amantadine ring, which increases blood plasma stability [6].
The Malaria tab on this ligand page provides additional curator comments of relevance to the Guide to MALARIA PHARMACOLOGY. |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Artefenomel has a longer elimination half-life than other antimalarial endoperoxides and therefore has potential as a single-dose treatment when used with a partner drug. A Phase 2 clinical trial in which artefenomel will be used in combination with ferroquine (NCT02497612) has commenced recruitment. Results from a trial with piperaquine (NCT02083380, published in [4]) did not reach target efficacy as a single-dose treatment and a trial in combination with DSM265 (NCT02573857) has been terminated. Click here to view the full list of artefenomel trials registered with ClinicalTrials.gov. |
Mechanism Of Action and Pharmacodynamic Effects  |
| The ozonide class are thought to act on the Plasmodium proteostatic stress response: initially disrupting haemoglobin metabolism and following extended exposure, to reflect clinically-relevant treatment periods, additional parasite biochemical pathways were found to be perturbed [3]. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT02083380 | Phase II Efficacy Study of Artefenomel & Piperaquine in Adults & Children With P. Falciparum Malaria. | Phase 2/Phase 3 Interventional | Medicines for Malaria Venture | ||
| NCT02497612 | To Evaluate the Efficacy of a Single Dose Regimen of Ferroquine and Artefenomel in Adults and Children With Uncomplicated Plasmodium Falciparum Malaria | Phase 2 Interventional | Sanofi | ||
| NCT02573857 | A Study to Characterise the Antimalarial and Transmission Blocking Activity of a Single Dose of DSM265 or OZ439 in Healthy Subjects With Induced Blood Stage Plasmodium Falciparum or Plasmodium Vivax Infection | Phase 1/Phase 2 Interventional | Medicines for Malaria Venture | ||
Pharmacokinetics  |
| Elimination |
| The estimated terminal half-life for elimination is 46 to 62 hours [5]. |