GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
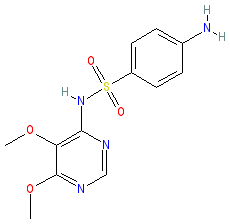
Synonyms: sulphadoxine
sulfadoxine is an approved drug (FDA (1981) in combination with pyrimethamine)
Compound class:
Synthetic organic
Comment: Sulfadoxine is a sulfonamide containing compound with antimalarial activity. It is used in the treatment of malaria as a combination therapy.
The Malaria tab on this ligand page provides additional curator comments of relevance to the Guide to MALARIA PHARMACOLOGY. Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| The 1981 US FDA approval for this compound is in combination with pyrimethamine (trade name Fansidar®). Sulfadoxine is listed in the World Health Organisation's 21st Essential Medicines List (2019) as a curative treatment for uncomplicated P. falciparum malaria in combination with pyrimethamine and artesunate. This is one of the artemisinin-based combination therapies (ACTs) recommended in the WHO's Guidelines for the treatment of malaria [2]. The link to extended ADME data provided below is for the combined formulation with pyrimethamine. |
External links  |
|
For extended ADME data see the following: Drugs.com |