GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Compound class:
Synthetic organic
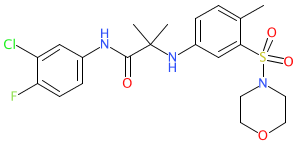
Comment: Compound 28 is the optimized lead from a novel class of antimalarial compounds based on an aminoacetamide scaffold [3].
The Malaria tab on this ligand page provides additional curator comments of relevance to the Guide to MALARIA PHARMACOLOGY. |
|
|||||||||||||||||||||||||||||||||||
| Guide to Malaria Pharmacology Comments |
| Compound selection from GlaxoSmithKline's Tres Cantos antimalarial set (TCAMS) [1-2] was used as the chemical starting point for the identification and optimization of a series of potent P. falciparum inhibitors with an aminoacetamide core. Although this class of compounds is not suitable for progression to pharmacokinetic and efficacy studies, due to metabolic instability and poor aqueous solubility, compound 28 has potential as a lead for further optimization or as a chemical tool for drug target identification [3]. Potential Target/Mechanism Of Action: As the precise mechanism of action of compound 28 is not yet known, we do not have a molecular target for this compound. |