GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
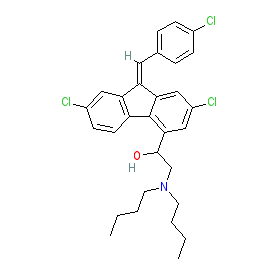
Synonyms: benflumetol
lumefantrine is an approved drug (FDA (2009) in combination with artemether)
Compound class:
Synthetic organic
Comment: Lumefantrine belongs to the aryl amino alcohols, a chemical class of antimalarial compounds that includes quinine, mefloquine and halofantrine.
Lumefantrine is a racemic mixture, with PubChem listing 9 stereoisotopes. The Malaria tab on this ligand page provides additional curator comments of relevance to the Guide to MALARIA PHARMACOLOGY. |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| The 2009 US FDA approval for this compound is in combination with artemether (trade name Coartem®) and is one of the artemisinin-based combination therapies (ACTs) recommended in the World Health Organisation's Guidelines for the treatment of malaria [5]. The European Medicines Agency (EMA) granted orphan designation for the combined formulation in 2010 (link to the EMA Orphan Designation record here). The link to extended ADME data provided below is for the combined formulation with artemether. Recruitment has commenced for a Phase 2 trial for combination therapy of lumefantrine with ganaplacide (NCT03167242). |
Mechanism Of Action and Pharmacodynamic Effects  |
| Although the mechanism of action in the Plasmodium parasite has not been fully elucidated, studies suggest that lumefantrine inhibits nucleic acid and protein synthesis [1]. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT03167242 | Efficacy and Safety of KAF156 in Combination With LUM-SDF in Adults and Children With Uncomplicated Plasmodium Falciparum Malaria | Phase 2 Interventional | Novartis | ||
Pharmacokinetics  |
| Elimination |
| The terminal elimination half-life of lumefantrine is 4.5 days [4]. |
External links  |
|
For extended ADME data see the following: Drugs.com |