GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
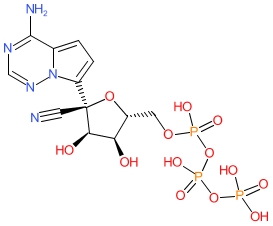
Synonyms: GS-441524 triphosphate | GS-443902 | RDV-TP
Compound class:
Synthetic organic
Comment: Remdesivir triphosphate (GS-443902) is a metabolite of remdesivir. It is formed by replacement of the 5'-hydroxyl group of GS-441524 by a triphosphate group. It inhibits RNA-dependent RNA polymerase (RdRP) activity in a range of viruses.
|
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Siegel D, Hui HC, Doerffler E, Clarke MO, Chun K, Zhang L, Neville S, Carra E, Lew W, Ross B et al.. (2017)
Discovery and Synthesis of a Phosphoramidate Prodrug of a Pyrrolo[2,1-f][triazin-4-amino] Adenine C-Nucleoside (GS-5734) for the Treatment of Ebola and Emerging Viruses. J Med Chem, 60 (5): 1648-1661. [PMID:28124907] |