GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Synonyms: SSR97193
Compound class:
Synthetic organic
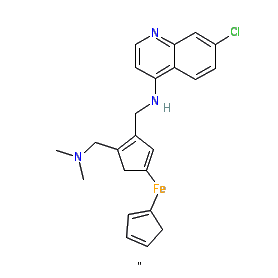
Comment: Ferroquine is a 4-aminoquinoline antimalarial compound with a novel ferrocenic group in its side chain.
The ferrocenic group causes rendering problems, with different images of the chemical structure found across web resources. We show the structure provided by NIH US National Library of Medicine (InChIKey IRJOHJGUSPNBRI-UHFFFAOYSA-N). PubChem provide a different InChIKey DLYPREQTTOHKSM-UHFFFAOYSA-N with two structural representations PubChem CID 56841801 (link in table below) and PubChem CID 76419023. The Malaria tab on this ligand page provides additional curator comments of relevance to the Guide to MALARIA PHARMACOLOGY. |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Biot C, Glorian G, Maciejewski LA, Brocard JS. (1997)
Synthesis and antimalarial activity in vitro and in vivo of a new ferrocene-chloroquine analogue. J Med Chem, 40 (23): 3715-8. [PMID:9371235] |
|
2. Biot C, Taramelli D, Forfar-Bares I, Maciejewski LA, Boyce M, Nowogrocki G, Brocard JS, Basilico N, Olliaro P, Egan TJ. (2005)
Insights into the mechanism of action of ferroquine. Relationship between physicochemical properties and antiplasmodial activity. Mol Pharm, 2 (3): 185-93. [PMID:15934779] |