|
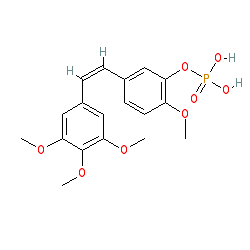
Synonyms: CA4DP | combretastatin A4 phosphate | fosbretabulin disodium | fosbretabulin tromethamine | Zybrestat (fosbretabulin disodium)
Compound class:
Synthetic organic
Comment: Fosbretabulin is being investigated for anti-tumour potential. It interferes with vascularisation by preventing tubulin-polymerisation and disrupting cell junctions [2]. This action destroys tumour vasculature and leads to cancer cell death and necrosis. Mechanistically, fosbretabulin is dephosphorylated to the active tubulin polymerisation inhibitor combretastatin A4 [3]. The compound was originally isolated from the African bush willow (Combretum caffrum) [1,5].
|
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Cirla A, Mann J. (2003)
Combretastatins: from natural products to drug discovery. Nat Prod Rep, 20 (6): 558-64. [PMID:14700199] |
|
2. Kanthou C, Tozer GM. (2009)
Microtubule depolymerizing vascular disrupting agents: novel therapeutic agents for oncology and other pathologies. Int J Exp Pathol, 90 (3): 284-94. [PMID:19563611] |
|
3. Lin CM, Singh SB, Chu PS, Dempcy RO, Schmidt JM, Pettit GR, Hamel E. (1988)
Interactions of tubulin with potent natural and synthetic analogs of the antimitotic agent combretastatin: a structure-activity study. Mol Pharmacol, 34 (2): 200-8. [PMID:3412321] |
|
4. Monk BJ, Sill MW, Walker JL, Darus CJ, Sutton G, Tewari KS, Martin LP, Schilder JM, Coleman RL, Balkissoon J et al.. (2016)
Randomized Phase II Evaluation of Bevacizumab Versus Bevacizumab Plus Fosbretabulin in Recurrent Ovarian, Tubal, or Peritoneal Carcinoma: An NRG Oncology/Gynecologic Oncology Group Study. J Clin Oncol, 34 (19): 2279-86. [PMID:27217446] |
|
5. Pettit GR, Singh SB, Hamel E, Lin CM, Alberts DS, Garcia-Kendall D. (1989)
Isolation and structure of the strong cell growth and tubulin inhibitor combretastatin A-4. Experientia, 45 (2): 209-11. [PMID:2920809] |