GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
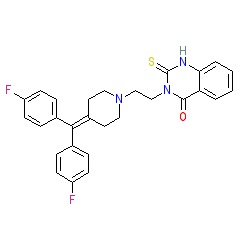
Synonyms: R 59 949 | R59949
Compound class:
Synthetic organic
Comment: This compound inhibits diacylglycerol kinase activity [2].
|
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Anastassiadis T, Deacon SW, Devarajan K, Ma H, Peterson JR. (2011)
Comprehensive assay of kinase catalytic activity reveals features of kinase inhibitor selectivity. Nat Biotechnol, 29 (11): 1039-45. [PMID:22037377] |
|
2. de Chaffoy de Courcelles D, Roevens P, Van Belle H, Kennis L, Somers Y, De Clerck F. (1989)
The role of endogenously formed diacylglycerol in the propagation and termination of platelet activation. A biochemical and functional analysis using the novel diacylglycerol kinase inhibitor, R 59 949. J Biol Chem, 264 (6): 3274-85. [PMID:2536741] |
|
3. Gao Y, Davies SP, Augustin M, Woodward A, Patel UA, Kovelman R, Harvey KJ. (2013)
A broad activity screen in support of a chemogenomic map for kinase signalling research and drug discovery. Biochem J, 451 (2): 313-28. [PMID:23398362] |
|
4. Jiang Y, Sakane F, Kanoh H, Walsh JP. (2000)
Selectivity of the diacylglycerol kinase inhibitor 3-[2-(4-[bis-(4-fluorophenyl)methylene]-1-piperidinyl)ethyl]-2, 3-dihydro-2-thioxo-4(1H)quinazolinone (R59949) among diacylglycerol kinase subtypes. Biochem Pharmacol, 59 (7): 763-72. [PMID:10718334] |