GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
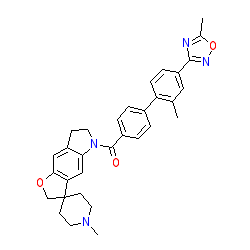
Synonyms: SB-224289 | SB224289
Compound class:
Synthetic organic
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Gaster LM, Blaney FE, Davies S, Duckworth DM, Ham P, Jenkins S, Jennings AJ, Joiner GF, King FD, Mulholland KR et al.. (1998)
The selective 5-HT1B receptor inverse agonist 1'-methyl-5-[[2'-methyl-4'-(5-methyl-1,2, 4-oxadiazol-3-yl)biphenyl-4-yl]carbonyl]-2,3,6,7-tetrahydro- spiro[furo[2,3-f]indole-3,4'-piperidine] (SB-224289) potently blocks terminal 5-HT autoreceptor function both in vitro and in vivo. J Med Chem, 41 (8): 1218-35. [PMID:9548813] |
|
2. Knight AR, Misra A, Quirk K, Benwell K, Revell D, Kennett G, Bickerdike M. (2004)
Pharmacological characterisation of the agonist radioligand binding site of 5-HT(2A), 5-HT(2B) and 5-HT(2C) receptors. Naunyn Schmiedebergs Arch Pharmacol, 370 (2): 114-23. [PMID:15322733] |
|
3. Newman-Tancredi A, Audinot V, Moreira C, Verrièle L, Millan MJ. (2000)
Inverse agonism and constitutive activity as functional correlates of serotonin h5-HT(1B) receptor/G-protein stoichiometry. Mol Pharmacol, 58 (5): 1042-9. [PMID:11040052] |
|
4. Selkirk JV, Scott C, Ho M, Burton MJ, Watson J, Gaster LM, Collin L, Jones BJ, Middlemiss DN, Price GW. (1998)
SB-224289--a novel selective (human) 5-HT1B receptor antagonist with negative intrinsic activity. Br J Pharmacol, 125 (1): 202-8. [PMID:9776361] |