|
Synonyms: 1263W94 | benzimidavir | Camvia® | G1263 | GW-1263 | Livtencity®
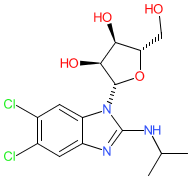
maribavir is an approved drug (FDA (2021), EMA (2022))
Compound class:
Synthetic organic
Comment: Maribavir is an orally bioavailable benzimidazole L-riboside that inhibits cytomegalovirus (CMV) pUL97 kinase with nM potency in vitro [1]. It inhibits viral replication.
|
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Biron KK, Harvey RJ, Chamberlain SC, Good SS, Smith 3rd AA, Davis MG, Talarico CL, Miller WH, Ferris R, Dornsife RE et al.. (2002)
Potent and selective inhibition of human cytomegalovirus replication by 1263W94, a benzimidazole L-riboside with a unique mode of action. Antimicrob Agents Chemother, 46 (8): 2365-72. [PMID:12121906] |
|
2. Kang C. (2022)
Maribavir: First Approval. Drugs, 82 (3): 335-340. [PMID:35147913] |
|
3. Marty FM, Ljungman P, Papanicolaou GA, Winston DJ, Chemaly RF, Strasfeld L, Young JA, Rodriguez T, Maertens J, Schmitt M et al.. (2011)
Maribavir prophylaxis for prevention of cytomegalovirus disease in recipients of allogeneic stem-cell transplants: a phase 3, double-blind, placebo-controlled, randomised trial. Lancet Infect Dis, 11 (4): 284-92. [PMID:21414843] |
|
4. Papanicolaou GA, Silveira FP, Langston AA, Pereira MR, Avery RK, Uknis M, Wijatyk A, Wu J, Boeckh M, Marty FM et al.. (2019)
Maribavir for Refractory or Resistant Cytomegalovirus Infections in Hematopoietic-cell or Solid-organ Transplant Recipients: A Randomized, Dose-ranging, Double-blind, Phase 2 Study. Clin Infect Dis, 68 (8): 1255-1264. [PMID:30329038] |