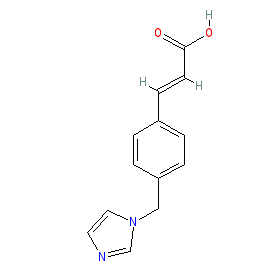
|
Synonyms: Cataclot® | KCT-0809 | OKY-046 | Pulmoza®
ozagrel is an approved drug (China and Japan only)
Compound class:
Synthetic organic
Comment: Ozagrel is a thromboxane A synthetase (CYP5A1; TBXAS1) inhibitor that has anti-inflammatory action [3]. Note that some of the bioactivity data for ozagrel relate to use of ozagrel sodium or ozagrel hydrochloride.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Ozagrel is used in China and Japan, for the treatment of ischemic cerebral disorders. This drug is not approved by the USA FDA or EMA. An opthalmic solution of ozagrel sodium (KCT-0809) reached Phase 3 clinical evaluation as a potential treatment for dry eye syndrome as experienced by patients with Sjögren's syndrome (NCT02503176), however the developer discontinued development in this indication and the study was terminated before completion. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT02503176 | An Extension Study of KCT-0809 in Dry Eye Patients With Sjögren's Syndrome | Phase 3 Interventional | Kissei Pharmaceutical Co., Ltd. | ||