GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
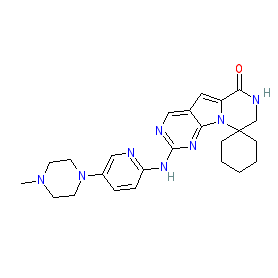
Synonyms: Cosela® | G1T28
trilaciclib is an approved drug (FDA (2021))
Compound class:
Synthetic organic
Comment: Trilaciclib (G1T28) is a short-acting CDK4/6 inhibitor that was developed by G1 Therapeutics [2].
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Pre-chemotherapy intravenous administration of G1T28 was evaluated in a number of clinical trials, in patients with advanced solid tumours- small cell lung cancer (NCT03041311) and triple-negative breast cancer (NCT02978716). It was approved by the FDA in February 2021, as a myelopreservation therapy indicated for patients receiving chemotherapy for small cell lung cancer. |
Mechanism Of Action and Pharmacodynamic Effects  |
| G1T28 represents a novel strategy for targeting CDK4/6-independent tumours. By transiently inhibiting CDK4/6 in normal hematopoietic stem cells, and thereby protecting them from chemotherapy-induced exhaustion, it is predicted that this strategy will deliver better outcomes for cancer patients receiving chemotherapy (i.e. reduced chemotherapy-induced myelosuppression) [1]. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT02978716 | Trilaciclib (G1T28), a CDK 4/6 Inhibitor, in Combination With Gemcitabine and Carboplatin in Metastatic Triple Negative Breast Cancer (mTNBC) | Phase 2 Interventional | G1 Therapeutics, Inc. | ||
| NCT03041311 | Carboplatin, Etoposide, and Atezolizumab With or Without Trilaciclib (G1T28), a CDK 4/6 Inhibitor, in Extensive Stage Small Cell Lung Cancer (SCLC) | Phase 2 Interventional | G1 Therapeutics, Inc. | ||