GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
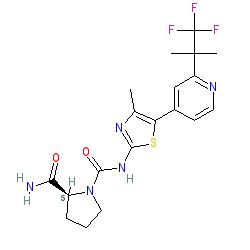
Synonyms: BYL 719 | BYL-719 | NVP-BYL719 | Piqray® | Vijoice®
alpelisib is an approved drug (FDA (2019),EMA (2020))
Compound class:
Synthetic organic
Comment: Alpelisib is an orally bioavailable phosphatidylinositol 3-kinase (PI3K) inhibitor that exhibits antineoplastic activity. It is selective for the α isoform (PIK3CA). It was the first PIK3CA inhibitor to be approved for the treatment of breast cancer.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Multiple clinical trials are assessing alpelisib in a range of PIK3CA mutated solid and haematologic cancers, either as a monotherapy or in combination with established and/or novel oncology drugs. Click here to link to ClinicalTrials.gov's full list of alpelisib trials. A combination therapy of alpelisib and fulvestrant was FDA approved in May 2019 [4], as a treatment for men and postmenopausal women with HR+ve, HER2-ve, PIK3CA-mutated advanced/metastatic breast cancer that has progressed on or after an endocrine-based therapy. CLOVES syndrome is a genetic disorder that results from gain-of-function mutations of the PIK3CA gene (e.g. the H1047R mutation), and is one of a spectrum of PIK3CA-related overgrowth syndromes (PROS). A study in France demonstrated that targeted therapy with alpelisib produced substantial therapeutic effects in all nineteen CLOVES patients included in the trial [5]. The EMA granted orphan designation as a PROS therapy in March 2021. In April 2022, the FDA approved alpelisib (Vijoice) as the first therapy for patients (≥2 years old) with severe manifestations of PROS. A case study in a child with congenital hyperinsulinism (due to homozygous deletion of ABCC8) found that adjuvant alpelisib improved glucose control without serious adverse effects, and deferred the necessity for total pancreatectomy [1]. |