GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
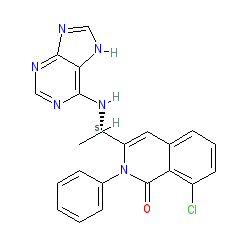
Synonyms: compound 4904 [Patent US8193182] [10] | Copiktra® | INK-1197 | IPI-145
duvelisib is an approved drug (FDA (2018), EMA (2021))
Compound class:
Synthetic organic
Comment: Duvelisib is an orally active, dual inhibitor of phosphoinositide-3 kinase (PI3K)-δ and PI3K-γ [14]. It was developed for the treatment of advanced hematolgical malignancies including CLL, SLL and FL [8,11-12].
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Early stage clinical efficacy was reported in hematological malignancies [3,6-7,9]. Duvelisib (under its long chemical name (S)-3-(1-(9H-purin-6-ylamino)ethyl)-8-chloro-2-phenylisoquinolin-1(2H)-one) was initially granted orphan drug (OD) designation by the EMA for the treatment of chronic lymphocytic leukemia (CLL) and by the FDA for CLL and follicular lymphoma. Full FDA approval was granted in September 2018 for the treatment of relapsed/refractory CLL/SLL after at least two prior therapies [5]. Accelerated approval for the treatment relapsed/refractory follicular lymphoma (FL) after at least two prior systemic therapies was also granted at this time. Duvelisib packaging carries a boxed warning to alert prescribers and patients to the risk of 4 fatal and/or serious toxicities that are associated with use of ths drug. These 4 adverse reactions are infections, diarrhea or colitis, cutaneous reactions, and pneumonitis. Click here to link to ClinicalTrials.gov's full list of duvelisib studies. In October 2019 FDA OD designation was granted for the treatment of T-cell lymphoma. The EMA has approved duvelisib (May 2021) as a treatment for CLL and follicular lymphoma |
Mechanism Of Action and Pharmacodynamic Effects  |
| Phosphoinositide-3 kinase (PI3K)-δ and PI3K-γ are preferentially expressed in immune cells. Inhibitors targeting these isozymes are hypothesized to modulate both the adaptive and innate immune responses, generating an anti-inflammatory effect. As the PI3Ks are essential mediators of the chemokine receptor signalling that is required for migration and survival of malignant hematological cells [1], inhibitors have been exploited as treatments for hematological cancers. Duvelisib has been shown to promote apoptosis in malignant B cells [2]. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT04372602 | Duvelisib to Combat COVID-19 | Phase 2 Interventional | Washington University School of Medicine | ||