GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
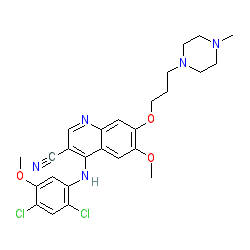
Synonyms: Bosulif® | SK 606 | SK-606 | SKI 606
bosutinib is an approved drug (FDA (2012), EMA (2013))
Compound class:
Synthetic organic
Comment: Bosutinib is a Type-1 kinase inhibitor. This compound is a dual inhibitor of Src family kinases and Abl kinase activity [9]. Bosutinib has been shown to stimulate macrophage clearance of bacterial infections [3]. Although it does not have intrinsic antibacterial activity, bosutinib could be used to boost host innate immunity and delivered as part of an antibacterial drug combination for the treatment for drug-resistant bacterial infections [3].
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖
View more information in the IUPHAR Pharmacology Education Project: bosutinib |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Bosutinib is approved for the treatment of adult patients with chronic, accelerated, or blast phase Philadelphia chromosome-positive (Ph+) chronic myelogenous leukemia (CML) with resistance, or intolerance to prior therapy. In December 2017, the FDA expanded approval to include treatment of newly-diagnosed chronic phase Ph+ CML [5]. Marketed formulations contain bosutinib monohydrate (PubChem CID 11990828). |
External links  |
|
For extended ADME data see the following: Electronic Medicines Compendium (eMC) Drugs.com European Medicines Agency (EMA) |