|
Synonyms: AB-1010 | AB1010 | Masican® (proposed trade name)
Compound class:
Synthetic organic
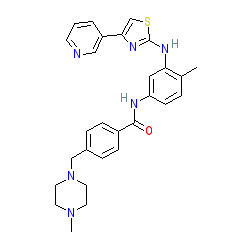
Comment: Using the trade name Masivet, this compound has been used for several years to treat mast cell tumours in dogs [6]. It is a receptor tyrosine kinase (RTK) inhibitor, with inhibitory activity at the cKIT, platelet derived growth factor receptor (PDGFR) and fibroblast growth factor receptor (FGFR) RTKs [4].
SARS-CoV-2: Using X-ray crystallography, masitinib has been shown to bind (non-covalently) into the catalytic site of the SARS-CoV-2 3CL protease (Mpro) [3], which corresponds with detection of in vitro Mpro inhibitory activity. It does not bind to the other viral protease PLpro. Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| The European Medicines Agency has granted masitinib orphan designation as a treatment for pancreatic cancer, amyotrophic lateral sclerosis (ALS, a.k.a. motor neurone disease, MND) and mastocytosis. It has been evaluated in clinical trials for a number of inflammatory conditions, solid tumours, multiple myeloma, ALS and Alzheimer's disease. Click here to link to ClinicalTrials.gov's complete listing of masitinib trials (note that many of these are not active or have been terminated). In June 2021, a Phase 3 study testing the safety and effectiveness of masitinib as an add-on treatment to riluzole for ALS was announced by AB Science. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT02588677 | Masitinib in Combination With Riluzole for the Treatment of Patients Suffering From Amyotrophic Lateral Sclerosis (ALS) | Phase 2/Phase 3 Interventional | AB Science | ||
| NCT03127267 | Efficacy and Safety of Masitinib Versus Placebo in the Treatment of ALS Patients | Phase 3 Interventional | AB Science | ||