GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
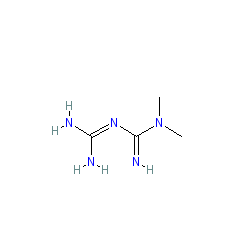
Synonyms: Glucophage® | LA-6023
metformin is an approved drug (FDA (1995))
Compound class:
Synthetic organic
Comment: Marketed formulations contain metformin hydrochloride (PubChem CID 14219). Metformin is often given in fixed-dose combinations with other antihyperglycemic agents.
Repurposing: Cancer cells undergo a metabolic switch to aerobic glycolysis, and become reliant on this metabolic pathway for energy (the Warburg effect). Inhibition of the glycolytic pathway is therefore considered as a tractable therapeutic target in oncology. As metformin is an inhibitor of glycolysis it is being examined for anti-cancer effects in a number of malignancies. Similarly, it is being examined for anti-inflammatory potential since activated immune cells also undergo a metabolic switch to aerobic glycolysis. If found to be effective, this could ultimately lead to metformin being repurposed for indications other than type 2 diabetes. 
View more information in the IUPHAR Pharmacology Education Project: metformin |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Used in the management of non-insulin dependent type 2 diabetes as an adjunct to diet and exercise. Also used in the management of polycystic ovary syndrome (PCOS). Metformin is combined with canagliflozin in Invokamet®, (also marketed as Vokanamet®) the first fixed-dose combination of an SGLT2 inhibitor with metformin to be approved (by the US FDA, 2014). Later in 2014, the US FDA approved a second SGLT2/metformin drug, Xigduo XR®, which contains dapagliflozin (as dapagliflozin propanediol monohydrate PubChem CID 56841155) and metformin hydrochloride. The first approval of a metformin-containing drug mixture by the EMA was for Avandamet (rosiglitazone plus metformin), although this drug was later withdrawn from the market. Avandamet has been superceeded by newer and more effective anti-diabetes combination drugs. |
Mechanism Of Action and Pharmacodynamic Effects  |
| Metformin works mainly by inhibiting glucose production and reducing its absorption in the gut, which appear to rely on metformin-induced activation of AMP-activated protein kinase (AMPK). |
External links  |
|
For extended ADME data see the following: Electronic Medicines Compendium (eMC) Drugs.com |