GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Synonyms: Lipostat® | Pravachol® | pravastatin sodium
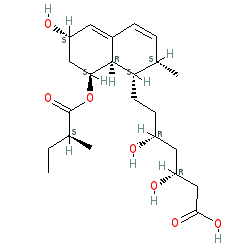
pravastatin is an approved drug (FDA (1991))
Compound class:
Synthetic organic
Comment: Pravastatin is a statin anti-dyslipidemia drug. Chemically the compound is a derivative of mevastatin (compactin), which was originally isolated from Penicillium citrinum.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖
View more information in the IUPHAR Pharmacology Education Project: pravastatin |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Used in the treatment of hypercholesterolemia and to reduce the risk of cardiovascular disease. The EMA approved a fixed-mixture drug (Pravafenix®) in 2011 which contains pravastatin and fenofibrate. |
External links  |
|
For extended ADME data see the following: Electronic Medicines Compendium (eMC) Drugs.com |