|
Synonyms: TL-895 | TL895
Compound class:
Synthetic organic
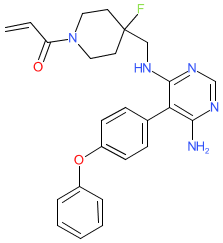
Comment: Example A225 [WO2012170976A2] is an investigational Bruton's tyrosine kinase (BTK) inhibitor. It is a fluoropiperidin analogue of evobrutinib and its chemical structure is claimed in Merck patent. This compound has been identified as TL-895 by online chemical resources, in which it is described as an orally-available, selective and irreversible BTK inhibitor. TL-895 was originally developed for potential to treat refractory myelofibrosis and acute myeloid leukemia (AML).
COVID-19: TL-895 has been redeployed for anti-inflammatory potential in COVID-19 patients. BTK inhibition is expected to reduce production of pro-inflammatory cytokines that are associated with cytokine storm, disease progression and poor prognosis in hospitalised COVID-19 patients. Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| TL-895 is being evaluated for immunosuppressive potential in myeloproliferative diseases and SARS-CoV-2 infection (COVID-19). |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT04878003 | Study of KRT-232 or TL-895 in Janus Associated Kinase Inhibitor Treatment-Naïve Myelofibrosis | Phase 2 Interventional | Kartos Therapeutics, Inc. | ||
| NCT04669067 | TL-895 and KRT-232 Study in Acute Myeloid Leukemia | Phase 1/Phase 2 Interventional | Telios Pharma, Inc. | ||
| NCT04419623 | A Study of TL-895 With Standard Available Treatment Versus Standard Available Treatment for the Treatment of COVID-19 in Patients With Cancer | Phase 1 Interventional | Telios Pharma, Inc. | ||