|
Synonyms: ERK-IN-1 | example 184 [WO2015066188A1] | LTT-462 | LTT462
Compound class:
Synthetic organic
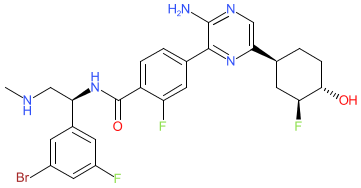
Comment: The chemical structure that was submitted to the WHO for INN rineterkib (LTT462), is claimed as example 184 in Novartis' kinase inhibitor patent WO2015066188A1 [1]. This structure maps to PubChem CID: 118045847 which provides an indication that it is an ERK inhibitor.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| LTT462 has been advanced to clinical evaluation in solid tumours. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT02711345 | A Phase I Clinical Study With Investigational Compound LTT462 in Adult Patients With Specific Advanced Cancers. | Phase 1 Interventional | Novartis | This study was not progressed to its dose expansion phase due to the limited clinical activity of LTT462. | |
| NCT04417621 | Study of Efficacy and Safety of LXH254 Combinations in Patients With Previously Treated Unresectable or Metastatic Melanoma | Phase 2 Interventional | Novartis | ||
| NCT04294160 | A Study of Select Drug Combinations in Adult Patients With Advanced/Metastatic BRAF V600 Colorectal Cancer | Phase 1 Interventional | Novartis | ||