GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Synonyms: AP24534 | Iclusig®
ponatinib is an approved drug (FDA (2012), EMA (2013))
Compound class:
Synthetic organic
Comment: Ponatinib is a Type-1 kinase inhibitor. It is a third generation BCR-Abl inhibitor. Use of ponatinib is subject to additional monitoring due to the observed serious risk of liver problems or blood clots (including heart attack and stroke, collectively referred to as arterial occlusive events, or AOEs). Final 5-year results of safety and efficacy in Ph+ leukemia as evaluated in NCT01207440 are reported by Cortes et al. (2018) [1].
Marketed formulations contain ponatinib hydrochloride (PubChem CID 46908927). Ponatinib has also been reported as a dual inhibitor of RIPK1 and RIPK3 which inhibits experimental models of RIPK1- and RIPK3-dependent cell death (necroptosis) [5]. On the basis of these findings, hybrid ponatinib/necrostatin-1 (a RIPK1 and IDO inhibitor) were designed and tested for potential to target RIPK1- and RIPK3-driven inflammatory pathologies. Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖
View more information in the IUPHAR Pharmacology Education Project: ponatinib |
|
|||||||||||||||||||||||||||||||||||
Classification  |
|
| Compound class | Synthetic organic |
| Approved drug? | Yes. US FDA (2012) | EU EMA (2013) |
IUPAC Name  |
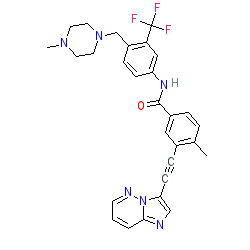
| 3-(2-imidazo[1,2-b]pyridazin-3-ylethynyl)-4-methyl-N-[4-[(4-methylpiperazin-1-yl)methyl]-3-(trifluoromethyl)phenyl]benzamide |
International Nonproprietary Names  |
|
| INN number | INN |
| 9362 | ponatinib |
Synonyms  |
| AP24534 | Iclusig® |
Database Links  |
|
| Specialist databases | |
Reactome Drug

|
R-ALL-9669717, R-ALL-9679854 |
Reactome Reaction

|
R-HSA-9669854, R-HSA-9669900, R-HSA-9674430, R-HSA-9674095, R-HSA-9702613, R-HSA-9679839, R-HSA-9693978, R-HSA-9702510, R-HSA-9695831 |
| Other databases | |
| CAS Registry No. | 943319-70-8 (source: SciFinder) |
| ChEBI | CHEBI:749637 |
| ChEMBL Ligand | CHEMBL1171837 |
| DrugCentral Ligand | 4716 |
| GtoPdb PubChem SID | 178102513 |
| PubChem CID | 24826799 |
| RCSB PDB Ligand | 0LI |
| Search Google for chemical match using the InChIKey | PHXJVRSECIGDHY-UHFFFAOYSA-N |
| Search Google for chemicals with the same backbone | PHXJVRSECIGDHY |
| Search PubMed clinical trials | ponatinib |
| Search PubMed titles | ponatinib |
| Search PubMed titles/abstracts | ponatinib |
| UniChem Compound Search for chemical match using the InChIKey | PHXJVRSECIGDHY-UHFFFAOYSA-N |
| UniChem Connectivity Search for chemical match using the InChIKey | PHXJVRSECIGDHY-UHFFFAOYSA-N |
| Wikipedia | Ponatinib |
Product suppliers
View disclaimer
Sponsored products disclaimer
Product supplier links are provided as a service to assist in identifying commercial suppliers of reagents that are mentioned on the IUPHAR/BPS Guide to PHARMACOLOGY database website, and do not imply their endorsement by NC-IUPHAR.
Links are provided in return for sponsorship, used to fund improvements to this database. The sponsorship account is managed and audited by the University of Edinburgh, a charitable body registered in Scotland, SC005336. If you are interested in sponsoring the database, please contact us.
✖
Sponsored products disclaimer
Product supplier links are provided as a service to assist in identifying commercial suppliers of reagents that are mentioned on the IUPHAR/BPS Guide to PHARMACOLOGY database website, and do not imply their endorsement by NC-IUPHAR.
Links are provided in return for sponsorship, used to fund improvements to this database. The sponsorship account is managed and audited by the University of Edinburgh, a charitable body registered in Scotland, SC005336. If you are interested in sponsoring the database, please contact us.
✖|
Ponatinib (links to external site)
Cat. No. HY-12047 |