GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
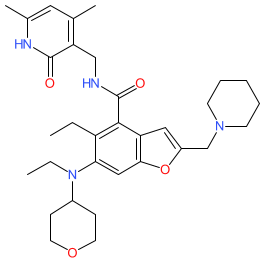
Synonyms: Airijing® (China) | compound 2 [WO2017084494] | compound Ia [WO2019091450]
zeprumetostat is an approved drug
Compound class:
Synthetic organic
Comment: The chemical structure for zeprumetostat was obtained from WHO proposed INN list 131 (August 2024). The INN record describes the compound as an enhancer of zeste homolog 2 (EZH2) inhibitor and antineoplastic. The chemical structure is claimed in patent WO2017084494A1 [3]. Based on Hengrui's declared development pipeline, we predicted at that time that zeprumetostat was likely the INN for their EZH2 inhibitor clinical lead SHR2554.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Zeprumetostat (SHR2554) was advanced as a clinical candidate antineoplastic for advanced lymphoid neoplasms [4]. Conditional (and first) approval was granted by China's NMPA in August 2025 [1-2], to treat relapsed/refractory peripheral T-cell lymphoma following ≥1 line of systemic therapy. Full approval will require satisfactory results from confirmatory clinical studies. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT06122389 | SHR2554 Clinical Study of Chidamide in the Treatment of T-cell Lymphoma | Phase 3 Interventional | Jiangsu HengRui Medicine Co., Ltd. | ||
| NCT06368167 | A Phase II Study of SHR2554 in Patients With Relapsed or Refractory Follicular Lymphoma | Phase 2 Interventional | Jiangsu HengRui Medicine Co., Ltd. | ||
| NCT03603951 | A Phase 1 Study of SHR2554 in Subjects With Relapsed or Refractory Mature Lymphoid Neoplasms | Phase 1 Interventional | Jiangsu HengRui Medicine Co., Ltd. | 5 | |