GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
thymidylate synthetase
Target id: 2642
Nomenclature: thymidylate synthetase
Abbreviated Name: TYMS
Family: 2.1.1.- Methyltransferases, Nucleoside synthesis and metabolism
Contents:
Gene and Protein Information  |
||||||
| Species | TM | AA | Chromosomal Location | Gene Symbol | Gene Name | Reference |
| Human | - | 313 | 18p11.32 | TYMS | thymidylate synthetase | |
| Mouse | - | 307 | 5 15.81 cM | Tyms | thymidylate synthase | |
| Rat | - | 307 | 9q38 | Tyms | thymidylate synthetase | |
Previous and Unofficial Names  |
| TMS | Tsase |
Database Links  |
|
| Alphafold | P04818 (Hs), P07607 (Mm), P45352 (Rn) |
| BRENDA | 2.1.1.45 |
| CATH/Gene3D | 3.30.572.10 |
| ChEMBL Target | CHEMBL1952 (Hs), CHEMBL3160 (Mm), CHEMBL4341 (Rn) |
| DrugBank Target | P04818 (Hs) |
| Ensembl Gene | ENSG00000176890 (Hs), ENSMUSG00000025747 (Mm), ENSRNOG00000037225 (Rn) |
| Entrez Gene | 7298 (Hs), 22171 (Mm), 29261 (Rn) |
| Human Protein Atlas | ENSG00000176890 (Hs) |
| KEGG Enzyme | 2.1.1.45 |
| KEGG Gene | hsa:7298 (Hs), mmu:22171 (Mm), rno:29261 (Rn) |
| OMIM | 188350 (Hs) |
| Pharos | P04818 (Hs) |
| RefSeq Nucleotide | NM_001071 (Hs), NM_021288 (Mm), NM_019179 (Rn) |
| RefSeq Protein | NP_001062 (Hs), NP_067263 (Mm), NP_062052 (Rn) |
| SynPHARM |
79619 (in complex with raltitrexed) 79620 (in complex with raltitrexed) |
| UniProtKB | P04818 (Hs), P07607 (Mm), P45352 (Rn) |
| Wikipedia | TYMS (Hs) |
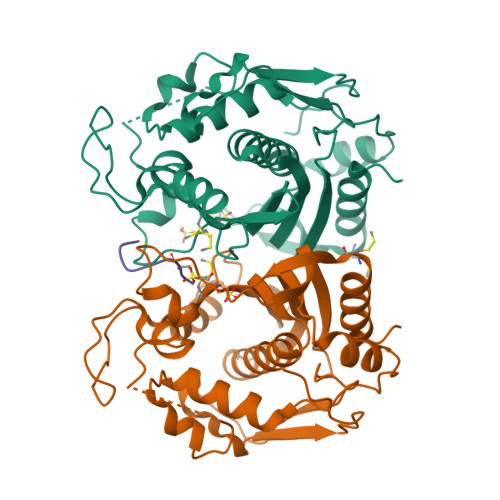
Selected 3D Structures  |
|||||||||||
|
|
||||||||||
Enzyme Reaction  |
||||
|
||||
Download all structure-activity data for this target as a CSV file 
| Inhibitors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| View species-specific inhibitor tables | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immuno Process Associations | ||||||||||||
|
||||||||||||
References
1. Bisset GM, Pawelczak K, Jackman AL, Calvert AH, Hughes LR. (1992) Syntheses and thymidylate synthase inhibitory activity of the poly-gamma-glutamyl conjugates of N-[5-[N-(3,4-dihydro-2-methyl-4-oxoquinazolin-6-ylmethyl)-N-methylamino ]-2-thenoyl]-L-glutamic acid (ICI D1694) and other quinazoline antifolates. J Med Chem, 35 (5): 859-66. [PMID:1372358]
2. Cardinale D, Guaitoli G, Tondi D, Luciani R, Henrich S, Salo-Ahen OM, Ferrari S, Marverti G, Guerrieri D, Ligabue A et al.. (2011) Protein-protein interface-binding peptides inhibit the cancer therapy target human thymidylate synthase. Proc Natl Acad Sci USA, 108 (34): E542-9. [PMID:21795601]
3. Carlini LE, Meropol NJ, Bever J, Andria ML, Hill T, Gold P, Rogatko A, Wang H, Blanchard RL. (2005) UGT1A7 and UGT1A9 polymorphisms predict response and toxicity in colorectal cancer patients treated with capecitabine/irinotecan. Clin Cancer Res, 11 (3): 1226-36. [PMID:15709193]
4. Gangjee A, Zhao Y, Ihnat MA, Thorpe JE, Bailey-Downs LC, Kisliuk RL. (2012) Novel tricyclic indeno[2,1-d]pyrimidines with dual antiangiogenic and cytotoxic activities as potent antitumor agents. Bioorg Med Chem, 20 (14): 4217-25. [PMID:22739090]
5. Gibbs DD, Theti DS, Wood N, Green M, Raynaud F, Valenti M, Forster MD, Mitchell F, Bavetsias V, Henderson E et al.. (2005) BGC 945, a novel tumor-selective thymidylate synthase inhibitor targeted to alpha-folate receptor-overexpressing tumors. Cancer Res, 65 (24): 11721-8. [PMID:16357184]
6. Marsham PR, Wardleworth JM, Boyle FT, Hennequin LF, Kimbell R, Brown M, Jackman AL. (1999) Design and synthesis of potent non-polyglutamatable quinazoline antifolate thymidylate synthase inhibitors. J Med Chem, 42 (19): 3809-20. [PMID:10508430]
7. Papamichael D. (1999) The use of thymidylate synthase inhibitors in the treatment of advanced colorectal cancer: current status. Oncologist, 4 (6): 478-87. [PMID:10631692]
8. Shih C, Habeck LL, Mendelsohn LG, Chen VJ, Schultz RM. (1998) Multiple folate enzyme inhibition: mechanism of a novel pyrrolopyrimidine-based antifolate LY231514 (MTA). Adv Enzyme Regul, 38: 135-52. [PMID:9762351]
How to cite this page
Nucleoside synthesis and metabolism: thymidylate synthetase. Last modified on 28/11/2018. Accessed on 02/07/2025. IUPHAR/BPS Guide to PHARMACOLOGY, https://www.guidetomalariapharmacology.org/GRAC/ObjectDisplayForward?objectId=2642.