GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
Contents:
Gene and Protein Information  |
||||||
| Species | TM | AA | Chromosomal Location | Gene Symbol | Gene Name | Reference |
| Human | - | 256 | 19p13.3 | PRTN3 | proteinase 3 | |
| Mouse | - | 254 | 10 39.72 cM | Prtn3 | proteinase 3 | |
| Rat | - | 254 | 7q11 | Prtn3 | proteinase 3 | |
Previous and Unofficial Names  |
| ACPA | AGP7 | Wegener granulomatosis autoantigen | C-ANCA | MBT | myeloblastin | PR3 |
Database Links  |
|
| Specialist databases | |
| MEROPS | S01.134 (Hs) |
| Other databases | |
| Alphafold | P24158 (Hs), Q61096 (Mm) |
| BRENDA | 3.4.21.76 |
| ChEMBL Target | CHEMBL3900 (Hs), CHEMBL5384 (Mm) |
| Ensembl Gene | ENSG00000196415 (Hs), ENSMUSG00000057729 (Mm), ENSRNOG00000029814 (Rn) |
| Entrez Gene | 5657 (Hs), 19152 (Mm), 314615 (Rn) |
| Human Protein Atlas | ENSG00000196415 (Hs) |
| KEGG Enzyme | 3.4.21.76 |
| KEGG Gene | hsa:5657 (Hs), mmu:19152 (Mm), rno:314615 (Rn) |
| OMIM | 177020 (Hs) |
| Orphanet | ORPHA118087 (Hs) |
| Pharos | P24158 (Hs) |
| RefSeq Nucleotide | NM_002777 (Hs), NM_011178 (Mm), NM_001024264 (Rn) |
| RefSeq Protein | NP_002768 (Hs), NP_035308 (Mm), NP_001019435 (Rn) |
| UniProtKB | P24158 (Hs), Q61096 (Mm) |
| Wikipedia | PRTN3 (Hs) |
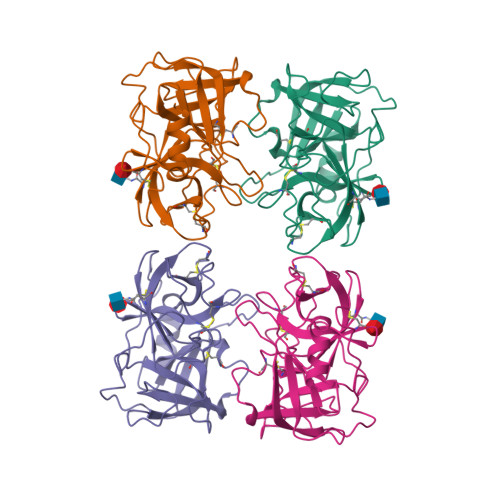
Selected 3D Structures  |
|||||||||||
|
|
||||||||||
Enzyme Reaction  |
||||
|
||||
Download all structure-activity data for this target as a CSV file 
| Inhibitors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunopharmacology Comments |
| Proteinase 3 (PR3), called myeloblastin when it was first identified, is an abundant serine protease found principally in neutrophil granules (but is also found on the surface of quiescent human neutrophils from peripheral blood). It is stored in the primary granules of circulating neutrophils alongside other cathepsin C-activated neutrophil serine proteases (NSPs), such as human neutrophil elastase (HNE), CatG, and NSP4. In pathological conditions it is thought that PR3 behaves to accelerate inflammation, by enhancing cytokine bioactivity, inactivating anti-inflammatory mediators and by promoting tissue injury (potentially by degrading extra-cellular matrix components like elastin, collagen, fibronectin, and laminins). In addition, imbalances between NSPs and their endogenous inhibitors can contribute towards pathological tissue damage, such as the damage associated with inflammatory lung diseases like chronic obstructive pulmonary disease (COPD), emphysema, and cystic fibrosis. PR3 inhibitors are considered to be useful clinical candidates for anti-inflammatory drug development [4]. |
| Cell Type Associations | ||||
|
Clinically-Relevant Mutations and Pathophysiology 
|
||||||||||||||
|
||||||||||||||
References
1. Epinette C, Croix C, Jaquillard L, Marchand-Adam S, Kellenberger C, Lalmanach G, Cadene M, Viaud-Massuard MC, Gauthier F, Korkmaz B. (2012) A selective reversible azapeptide inhibitor of human neutrophil proteinase 3 derived from a high affinity FRET substrate. Biochem Pharmacol, 83 (6): 788-96. [PMID:22209715]
2. Fujinaga M, Chernaia MM, Halenbeck R, Koths K, James MN. (1996) The crystal structure of PR3, a neutrophil serine proteinase antigen of Wegener's granulomatosis antibodies. J Mol Biol, 261 (2): 267-78. [PMID:8757293]
3. Guarino C, Gruba N, Grzywa R, Dyguda-Kazimierowicz E, Hamon Y, Łȩgowska M, Skoreński M, Dallet-Choisy S, Marchand-Adam S, Kellenberger C et al.. (2018) Exploiting the S4-S5 Specificity of Human Neutrophil Proteinase 3 to Improve the Potency of Peptidyl Di(chlorophenyl)-phosphonate Ester Inhibitors: A Kinetic and Molecular Modeling Analysis. J Med Chem, 61 (5): 1858-1870. [PMID:29442501]
4. Korkmaz B, Lesner A, Guarino C, Wysocka M, Kellenberger C, Watier H, Specks U, Gauthier F, Jenne DE. (2016) Inhibitors and Antibody Fragments as Potential Anti-Inflammatory Therapeutics Targeting Neutrophil Proteinase 3 in Human Disease. Pharmacol Rev, 68 (3): 603-30. [PMID:27329045]
5. Santana AB, Lucas SD, Gonçalves LM, Correia HF, Cardote TA, Guedes RC, Iley J, Moreira R. (2012) N-Acyl and N-sulfonyloxazolidine-2,4-diones are pseudo-irreversible inhibitors of serine proteases. Bioorg Med Chem Lett, 22 (12): 3993-7. [PMID:22595175]
How to cite this page
S1: Chymotrypsin: proteinase 3. Last modified on 27/02/2018. Accessed on 29/04/2026. IUPHAR/BPS Guide to PHARMACOLOGY, https://www.guidetomalariapharmacology.org/GRAC/ObjectDisplayForward?objectId=2401.