GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
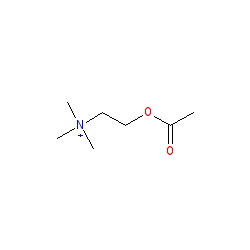
Synonyms: ACh | E1001 | O-acetylcholine
Compound class:
Metabolite
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖
View more information in the IUPHAR Pharmacology Education Project: acetylcholine |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Bravo DT, Kolmakova NG, Parsons SM. (2004)
Choline is transported by vesicular acetylcholine transporter. J Neurochem, 91 (3): 766-8. [PMID:15485505] |
|
2. Cheng K, Khurana S, Chen Y, Kennedy RH, Zimniak P, Raufman JP. (2002)
Lithocholylcholine, a bile acid/acetylcholine hybrid, is a muscarinic receptor antagonist. J Pharmacol Exp Ther, 303 (1): 29-35. [PMID:12235229] |
|
3. Jakubík J, Bacáková L, El-Fakahany EE, Tucek S. (1997)
Positive cooperativity of acetylcholine and other agonists with allosteric ligands on muscarinic acetylcholine receptors. Mol Pharmacol, 52 (1): 172-9. [PMID:9224827] |
|
4. Keov P, Valant C, Devine SM, Lane JR, Scammells PJ, Sexton PM, Christopoulos A. (2013)
Reverse engineering of the selective agonist TBPB unveils both orthosteric and allosteric modes of action at the M₁ muscarinic acetylcholine receptor. Mol Pharmacol, 84 (3): 425-37. [PMID:23798605] |
|
5. Khare P, Ojeda AM, Chandrasekaran A, Parsons SM. (2010)
Possible important pair of acidic residues in vesicular acetylcholine transporter. Biochemistry, 49 (14): 3049-59. [PMID:20225888] |
|
6. Kovacs I, Yamamura HI, Waite SL, Varga EV, Roeske WR. (1998)
Pharmacological comparison of the cloned human and rat M2 muscarinic receptor genes expressed in the murine fibroblast (B82) cell line. J Pharmacol Exp Ther, 284 (2): 500-7. [PMID:9454790] |