GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Compound class:
Synthetic organic
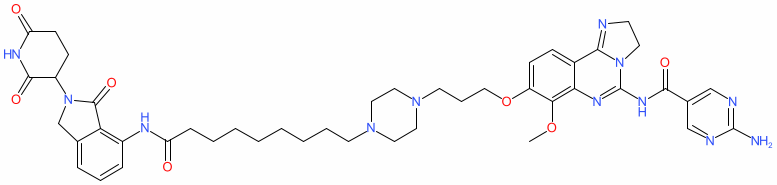
Comment: This is a heterobifunctional cereblon-binding PROTAC degrader that is selective for phosphoinositide 3-kinase (PI3K) isoforms α and δ [1]. It exploits copanlisib as the PI3K-targeting warhead. D5 is proposed for therapeutic potential in PIK3CA mutant breast cancers (with the oncogenic H1047R mutation for example).
|
|
|||||||||||||||||||||||||||||||||||
| Bioactivity Comments |
| DC50 for PI3Kα is 0.05 nM, whereas the DC50 values for the β and γ isozymes are >1 μM. |