GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
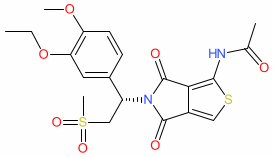
Synonyms: Hemay005 | Momishin® (China)
mufemilast is an approved drug
Compound class:
Synthetic organic
|
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Mufemilast (Hemay005) was advanced as an immunomodulatory clinical lead for a broad-spectrum of chronic inflammatory diseases. The FDA granted orphan drug designation for Behcet`s syndrome. First approval was granted by China's NMPA in 2025, to treat moderate to severe plaque psoriasis [2]. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT04839328 | A Phase Ⅲ Efficacy and Safety Study of Hemay005 in Subjects With Moderate to Severe Plaque Psoriasis | Phase 3 Interventional | Ganzhou Hemay Pharmaceutical Co., Ltd | ||
| NCT07314242 | A Study to Investigate the Biomarkers of Hemay005 in Adult Participants With Moderate to Severe COPD | Phase 2 Interventional | Ganzhou Hemay Pharmaceutical Co., Ltd | ||
| NCT06145893 | A Study of Efficacy and Safety of Hemay005 Tablets in Patients With Behçet's Disease | Phase 3 Interventional | Ganzhou Hemay Pharmaceutical Co., Ltd | ||
| NCT03007810 | Safety and Pharmacokinetics of Hemay005 In Healthy Male Subjects | Phase 1 Interventional | Tianjin Hemay Bio-Tech Co., Ltd | 3 | |