GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
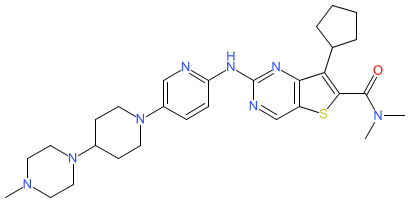
Synonyms: Example 5 [WO2017193872] | FCN-437 | FCN-437c | FCN437 | Futuoning® (China)
fovinaciclib is an approved drug
Compound class:
Synthetic organic
Comment: The chemical structure for fovinaciclib was obtained from proposed INN list 130 (Feb. 2024), in which the compound is described as cyclin dependent kinase (CDK) inhibitor with antineoplastic potential. This compound is claimed in patent WO2017193872A1 (Fochon Pharmaceutical) [2]. It appears to be CDK4/6 inhibitor selective inhibitor FCN-437/FCN-437c [1].
|
|
|||||||||||||||||||||||||||||||||||
| No information available. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT05004142 | Study of Efficacy, Safety, and Pharmacokinetics of FCN-437c in Combination With Fulvestrant or Letrozole+Goserelin | Phase 2 Interventional | Ahon Pharmaceutical Co., Ltd. | ||
| NCT05439499 | This is a Multicenter, Randomized, Double-blind, Placebo-controlled Phase III Clinical Study Evaluating the Efficacy and Safety of FCN-437c Versus Placebo in Combination With Letrozole or Anastrozole ± Goserelin in Women With HR+ and HER2- Advanced Breast Cancer. | Phase 3 Interventional | Ahon Pharmaceutical Co., Ltd. | ||
| NCT05438810 | This is a Multicenter, Randomized, Double-blind, Placebo-controlled Phase III Clinical Study Evaluating the Efficacy and Safety of FCN-437c in Combination With Fluvestrant ± Goseraline Versus Placebo Combined With Fulvestrant ± Goserelin in Women With HR+ and HER2- Advanced Breast Cancer. | Phase 3 Interventional | Ahon Pharmaceutical Co., Ltd. | ||