GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Synonyms: KBP-7072
Compound class:
Synthetic organic
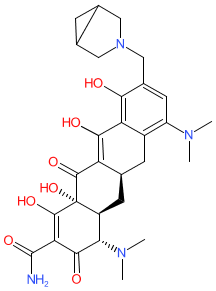
Comment: Zifanocycline is an aminomethylcycline (tetracycline derivative) with broad-spectrum antibacterial activity [3]. We show the chemical structure provided by PubChem, but note that this is not an exact match for the CAS number assigned in the WHO INN record for this drug.
|
|
|||||||||||||||||||||||||||||||||||
| Bioactivity Comments |
| Zifanocycline has potent in vitro activity against both Gram-positive and Gram-negative bacteria, including methicillin-resistant Staphylococcus aureus (MRSA) isolates (MIC90 of 0.12 μg/ml) and Haemophilus influenzae (MIC90 of 0.25 μg/ml) [3]. The MIC90 for in vitro antibacterial activity against Acinetobacter baumannii clinical isolates is 1μg/ml [1-2]. |