GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
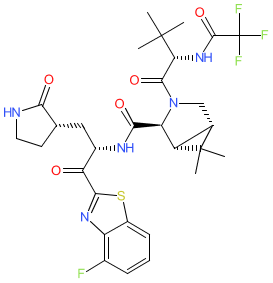
Synonyms: compound 5 [PMID: 37756225] | TKB-245
Compound class:
Synthetic organic
Comment: This compound is claimed in patent WO/2023/286844, as an inhibitor of coronavirus' Mpro [2]. Name to structure is also disclosed in [1] and [3].
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Higashi-Kuwata N, Tsuji K, Hayashi H, Bulut H, Kiso M, Imai M, Ogata-Aoki H, Ishii T, Kobayakawa T, Nakano K et al.. (2023)
Identification of SARS-CoV-2 Mpro inhibitors containing P1' 4-fluorobenzothiazole moiety highly active against SARS-CoV-2. Nat Commun, 14 (1): 1076. [PMID:36841831] |
|
2. Mitsuya H, Tamamura H, Kuwata N. (2023)
COMPOUND EXHIBITING PHYSIOLOGICAL ACTIVITY SUCH AS ANTIVIRAL ACTIVITY. Patent number: WO2023286844. Assignee: NATIONAL CENTER FOR GLOBAL HEALTH AND MEDICINE [JP]. Priority date: 15/07/2021. Publication date: 10/01/2023. |
|
3. Tsuji K, Ishii T, Kobayakawa T, Higashi-Kuwata N, Shinohara K, Azuma C, Miura Y, Nakano H, Wada N, Hattori SI et al.. (2023)
Structure-Activity Relationship Studies of SARS-CoV-2 Main Protease Inhibitors Containing 4-Fluorobenzothiazole-2-carbonyl Moieties. J Med Chem, 66 (19): 13516-13529. [PMID:37756225] |