GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
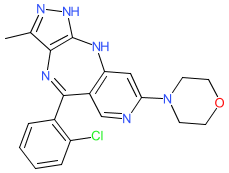
Synonyms: Example 6 [US10889586B2] | TT-00420 | TT00420
Compound class:
Synthetic organic
Comment: The chemical structure for tinengotinib was revealed in WHO proposed INN list 126 (Jan 2022), in which it was described as a tyrosine kinase inhibitor and antineoplastic agent. The SMILES provided a match to TransThera Sciences' clinical lead TT-00420 via PubChem. TT-00420 appears to be an orally administered multi‐kinase inhibitor that targets cell proliferation, angiogenesis, and immune-oncology, through inhibition of Aurora kinases A/B and Janus kinases (involved in cytokine signalling) and receptor tyrosine kinases such as FGFRs and VEGFRs (which play roles in proliferation and angiogenesis) [1].
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| TT-00420 has been advanced to clinical trials in patients with various advanced solid tumours. It was granted FDA Fast Track designation in November 2021. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT04705922 | Relative Bioavailability Study and Food Effect Study of TT-00420 Capsule and Tablet Formulations in Healthy Volunteers | Phase 1 Interventional | TransThera Sciences (Nanjing), Inc. | ||
| NCT03654547 | Safety of TT-00420 Monotherapy in Patients With Advanced Solid Tumors and Triple Negative Breast Cancer | Phase 1 Interventional | TransThera Sciences (Nanjing), Inc. | ||
| NCT04919642 | Study to Evaluate the Efficacy and Safety of TT-00420 in Cholangiocarcinoma | Phase 2 Interventional | TransThera Sciences (Nanjing), Inc. | ||
| NCT04742959 | Study of TT-00420 Tablet as Monotherapy and Combination Therapy in Patients With Advanced Solid Tumors | Phase 1/Phase 2 Interventional | TransThera Sciences (Nanjing), Inc. | ||