GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
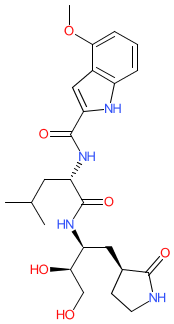
Compound class:
Synthetic organic
|
|
|||||||||||||||||||||||||||||||||||
| Bioactivity Comments |
| Example 2 inhibits 3CL protease (Mpro) of the SARS-CoV-1 coronavirus with an IC50 of 4 nM (in a FRET assay measuring protease catalysed cleavage of a TAMRA-labelled substrate peptide) and has in vitro antiviral activity [1]. |