GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
acetyl-CoA acetyltransferase 1
Target id: 2435
Nomenclature: acetyl-CoA acetyltransferase 1
Family: Lanosterol biosynthesis pathway
Contents:
Gene and Protein Information  |
||||||
| Species | TM | AA | Chromosomal Location | Gene Symbol | Gene Name | Reference |
| Human | - | 427 | 11q22.3 | ACAT1 | acetyl-CoA acetyltransferase 1 | |
| Mouse | - | 424 | 9 29.12 cM | Acat1 | acetyl-Coenzyme A acetyltransferase 1 | |
| Rat | - | 424 | 8q24 | Acat1 | acetyl-CoA acetyltransferase 1 | |
Previous and Unofficial Names  |
| ACAT | acetoacetyl-CoA thiolase | acetyl-Co A acetyltransferase 1 mitochondrial | acetoacetyl coenzyme A thiolase |
Database Links  |
|
| Alphafold | P24752 (Hs), Q8QZT1 (Mm), P17764 (Rn) |
| BRENDA | 2.3.1.9 |
| CATH/Gene3D | 3.40.47.10 |
| ChEMBL Target | CHEMBL2616 (Hs), CHEMBL3393 (Rn) |
| Ensembl Gene | ENSG00000075239 (Hs), ENSMUSG00000032047 (Mm), ENSRNOG00000007862 (Rn) |
| Entrez Gene | 38 (Hs), 110446 (Mm), 25014 (Rn) |
| Human Protein Atlas | ENSG00000075239 (Hs) |
| KEGG Enzyme | 2.3.1.9 |
| KEGG Gene | hsa:38 (Hs), mmu:110446 (Mm), rno:25014 (Rn) |
| OMIM | 607809 (Hs) |
| Orphanet | ORPHA117724 (Hs) |
| Pharos | P24752 (Hs) |
| RefSeq Nucleotide | NM_000019 (Hs), NM_144784 (Mm), NM_017075 (Rn) |
| RefSeq Protein | NP_000010 (Hs), NP_659033 (Mm), NP_058771 (Rn) |
| UniProtKB | P24752 (Hs), Q8QZT1 (Mm), P17764 (Rn) |
| Wikipedia | ACAT1 (Hs) |
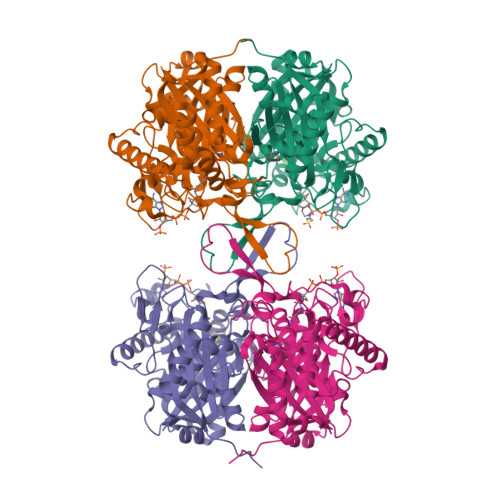
Selected 3D Structures  |
|||||||||||
|
|
||||||||||
Enzyme Reaction  |
||||
|
||||
Substrates and Reaction Kinetics  |
||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||
Download all structure-activity data for this target as a CSV file 
| Inhibitors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| View species-specific inhibitor tables | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Clinically-Relevant Mutations and Pathophysiology 
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
References
1. Ban H, Muraoka M, Ioriya K, Ohashi N. (2006) Synthesis and biological activity of novel 4-phenyl-1,8-naphthyridin-2(1H)-on-3-yl ureas: potent acyl-CoA:cholesterol acyltransferase inhibitor with improved aqueous solubility. Bioorg Med Chem Lett, 16 (1): 44-8. [PMID:16242323]
2. Fukao T, Nakamura H, Song XQ, Nakamura K, Orii KE, Kohno Y, Kano M, Yamaguchi S, Hashimoto T, Orii T et al.. (1998) Characterization of N93S, I312T, and A333P missense mutations in two Japanese families with mitochondrial acetoacetyl-CoA thiolase deficiency. Hum Mutat, 12 (4): 245-54. [PMID:9744475]
3. Fukao T, Yamaguchi S, Orii T, Schutgens RB, Osumi T, Hashimoto T. (1992) Identification of three mutant alleles of the gene for mitochondrial acetoacetyl-coenzyme A thiolase. A complete analysis of two generations of a family with 3-ketothiolase deficiency. J Clin Invest, 89 (2): 474-9. [PMID:1346617]
4. Fukao T, Yamaguchi S, Tomatsu S, Orii T, Frauendienst-Egger G, Schrod L, Osumi T, Hashimoto T. (1991) Evidence for a structural mutation (347Ala to Thr) in a German family with 3-ketothiolase deficiency. Biochem Biophys Res Commun, 179 (1): 124-9. [PMID:1715688]
5. Haapalainen AM, Meriläinen G, Pirilä PL, Kondo N, Fukao T, Wierenga RK. (2007) Crystallographic and kinetic studies of human mitochondrial acetoacetyl-CoA thiolase: the importance of potassium and chloride ions for its structure and function. Biochemistry, 46 (14): 4305-21. [PMID:17371050]
6. Ikenoya M, Yoshinaka Y, Kobayashi H, Kawamine K, Shibuya K, Sato F, Sawanobori K, Watanabe T, Miyazaki A. (2007) A selective ACAT-1 inhibitor, K-604, suppresses fatty streak lesions in fat-fed hamsters without affecting plasma cholesterol levels. Atherosclerosis, 191 (2): 290-7. [PMID:16820149]
7. Wakazono A, Fukao T, Yamaguchi S, Hori T, Orii T, Lambert M, Mitchell GA, Lee GW, Hashimoto T. (1995) Molecular, biochemical, and clinical characterization of mitochondrial acetoacetyl-coenzyme A thiolase deficiency in two further patients. Hum Mutat, 5 (1): 34-42. [PMID:7728148]
8. Wang X, Wang H, Cao M, Li Z, Chen X, Patenia C, Gore A, Abboud EB, Al-Rajhi AA, Lewis RA et al.. (2011) Whole-exome sequencing identifies ALMS1, IQCB1, CNGA3, and MYO7A mutations in patients with Leber congenital amaurosis. Hum Mutat, 32 (12): 1450-9. [PMID:21901789]