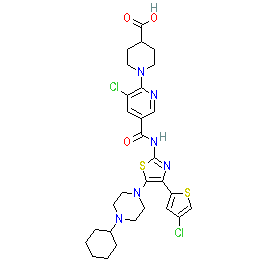
|
avatrombopag is an approved drug (FDA (2018), EMA (2019))
Compound class:
Synthetic organic
Comment: Avatrombopag is a second generation, small molecule, orally administered thrombopoietin receptor agonist [1]. It was discovered through its ability to mimic the action of endogenous thrombopoietin which is the primary physiological regulator of normal platelet production. The pharmacokinetics, pharmacodynamics, pharmacogenomics, safety, and tolerability of avatrombopag were reported by Nomoto et al. in 2017 [2].
|
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Fukushima-Shintani M, Suzuki K, Iwatsuki Y, Abe M, Sugasawa K, Hirayama F, Kawasaki T, Nakahata T. (2009)
AKR-501 (YM477) a novel orally-active thrombopoietin receptor agonist. Eur J Haematol, 82 (4): 247-54. [PMID:19183407] |
|
2. Nomoto M, Pastino G, Rege B, Aluri J, Ferry J, Han D. (2018)
Pharmacokinetics, Pharmacodynamics, Pharmacogenomics, Safety, and Tolerability of Avatrombopag in Healthy Japanese and White Subjects. Clin Pharmacol Drug Dev, 7 (2): 188-195. [PMID:28339166] |
|
3. Terrault N, Chen YC, Izumi N, Kayali Z, Mitrut P, Tak WY, Allen LF, Hassanein T. (2018)
Avatrombopag Before Procedures Reduces Need for Platelet Transfusion in Patients With Chronic Liver Disease and Thrombocytopenia. Gastroenterology, 155 (3): 705-718. [PMID:29778606] |
|
4. Terrault NA, Hassanein T, Howell CD, Joshi S, Lake J, Sher L, Vargas H, McIntosh J, Tang S, Jenkins TM. (2014)
Phase II study of avatrombopag in thrombocytopenic patients with cirrhosis undergoing an elective procedure. J Hepatol, 61 (6): 1253-9. [PMID:25048952] |