|
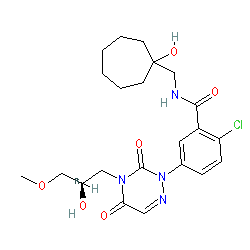
Synonyms: CE-224,535 | CE-224535 | compound 3 [PMID: 21782426] | compound 33 [PMID: 21565499]
Compound class:
Synthetic organic
Comment: PF-04905428 is an investigational purinergic receptor 2 (P2X7) antagonist [2,4], with potential antiinflammatory action in various conditions such as arthritis, allergies, asthma, COPD, autoimmune diseases and other disorders.. PF-04905428 is one of the compounds claimed in patent US6974812 [1].
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| A Phase 3 clinical trial in patients with rheumatoid arthritis (RA: NCT00628095) has been completed. Results showed PF-04905428 (CE-224,535) was not effective in treating RA [3]. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT00628095 | Study of CE-224,535 A Twice Daily Pill To Control Rheumatoid Arthritis In Patients Who Have Not Totally Improved With Methotrexate | Phase 2/Phase 3 Interventional | Pfizer | ||