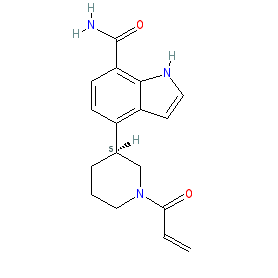
|
Synonyms: example #30 (S) [WO2014210255A1]
Compound class:
Synthetic organic
Comment: Elsubrutinib (example #30 (S) [WO2014210255A1]) is the (S) stereoenantiomer of example #30 from Abbvie's patent WO2014210255A1 which claims BTK inhibitors for their potential to treat immunological and oncological conditions [1]. Example #30 (S) could be ABBV-105, an Abbvie BTK inhibitor that is being evaluated in combination with upadacitinib (ABT-494, a JAK1 inhibitor) in patients with rheumatoid arthritis or systemic lupus erythematosus (the combination is coded ABBV-599). Although preclinical efficacy has been reported [2], the structure of ABBV-105 has not been formally disclosed, so the name-to-structure alluded to here is speculative and provided in good faith.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Elsubrutinib has progressed to Phase 2 evaluations to determine efficacy in trial subjects with rheumatoid arthritis or systemic lupus erythematosus. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT04451772 | A Study of the Safety of Oral Elsubrutinib Capsules and Oral Upadacitinib Tablets Given Alone or in Combination (ABBV-599) for Adult Participants With Moderately to Severely Active Systemic Lupus Erythematosus to Assess Change in Disease State | Phase 2 Interventional | AbbVie | ||
| NCT03978520 | A Study to Investigate the Safety and Efficacy of Elsubrutinib and Upadacitinib Given Alone or in Combination in Participants With Moderately to Severely Active Systemic Lupus Erythematosus (SLE) | Phase 2 Interventional | AbbVie | ||
| NCT03682705 | A Study to Investigate the Safety and Efficacy of ABBV-105 Alone or in Combination With Upadacitinib (ABBV-599 Combination) in Participants With Active Rheumatoid Arthritis | Phase 2 Interventional | AbbVie | ||